Quick filters:
Page 1 of 1
C2h5 Stock Photos and Images
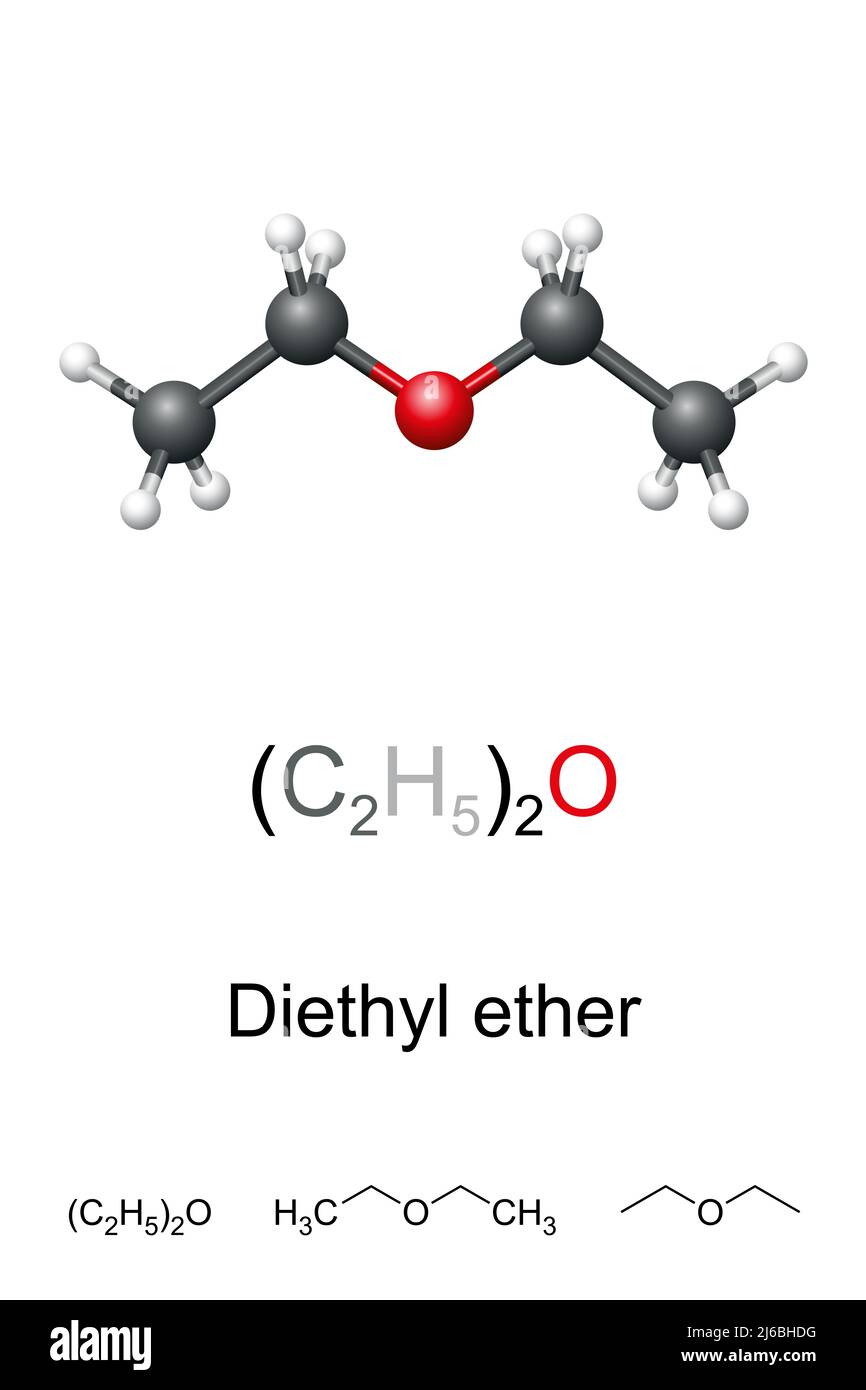 Diethyl ether, or simply ether, ball-and-stick model, molecular and chemical formula. Ethoxyethane, (C2H5)2O, an organic compound, also known as Et2O. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/diethyl-ether-or-simply-ether-ball-and-stick-model-molecular-and-chemical-formula-ethoxyethane-c2h52o-an-organic-compound-also-known-as-et2o-image468623052.html
Diethyl ether, or simply ether, ball-and-stick model, molecular and chemical formula. Ethoxyethane, (C2H5)2O, an organic compound, also known as Et2O. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/diethyl-ether-or-simply-ether-ball-and-stick-model-molecular-and-chemical-formula-ethoxyethane-c2h52o-an-organic-compound-also-known-as-et2o-image468623052.htmlRF2J6BHDG–Diethyl ether, or simply ether, ball-and-stick model, molecular and chemical formula. Ethoxyethane, (C2H5)2O, an organic compound, also known as Et2O.
 air is bubbled through ether in a beaker cooling water on the bench until it freezes Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-air-is-bubbled-through-ether-in-a-beaker-cooling-water-on-the-bench-18608055.html
air is bubbled through ether in a beaker cooling water on the bench until it freezes Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-air-is-bubbled-through-ether-in-a-beaker-cooling-water-on-the-bench-18608055.htmlRMB27JNB–air is bubbled through ether in a beaker cooling water on the bench until it freezes
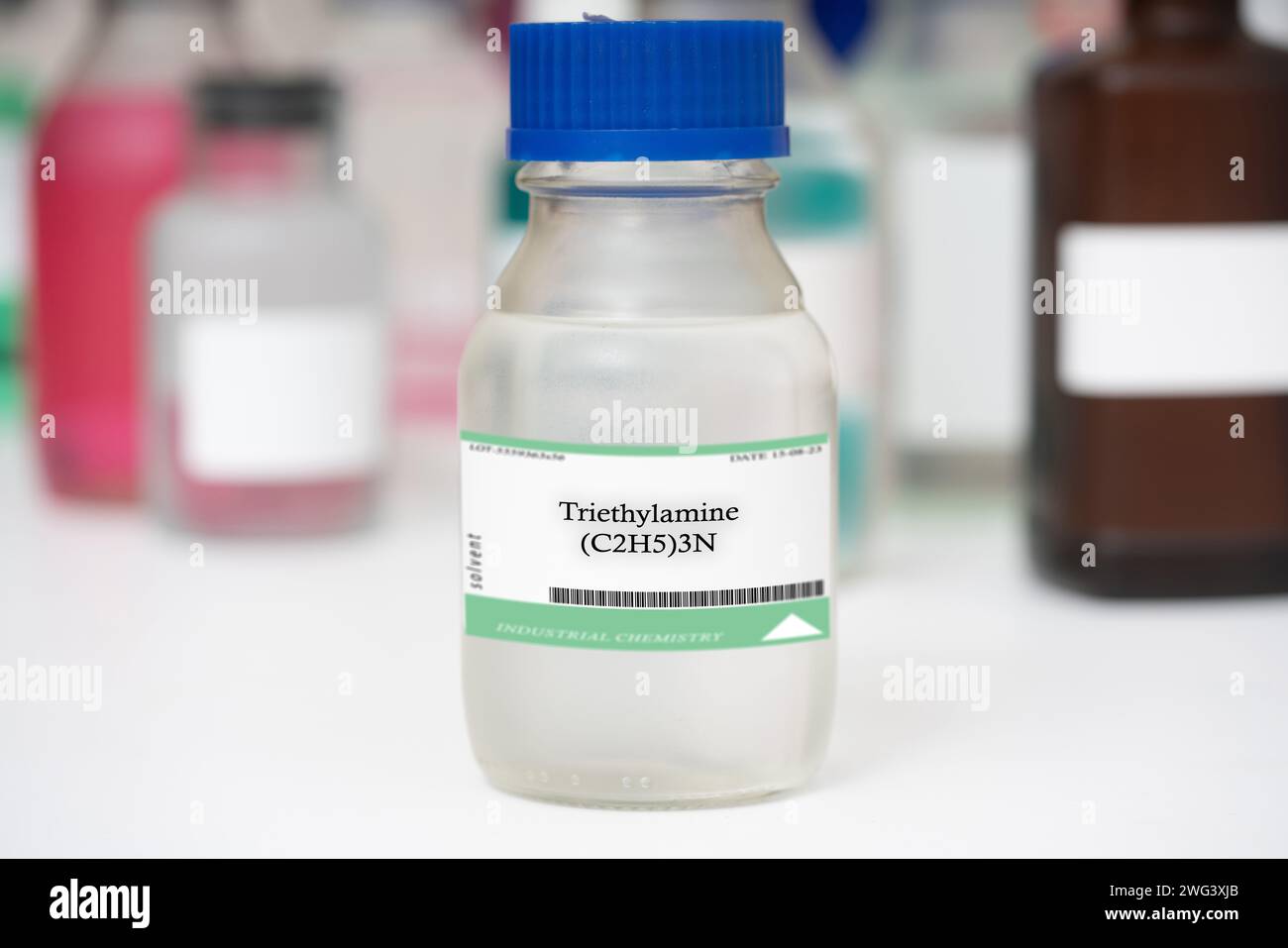 Bottle of triethylamine Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottle-of-triethylamine-image595073763.html
Bottle of triethylamine Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bottle-of-triethylamine-image595073763.htmlRF2WG3XJB–Bottle of triethylamine
 Chemistry : general, medical, and pharmaceutical including the chemistry of the U.Spharmacopoeia ; a manual on the general principles of the science, and their applications in medicine and pharmacy . due its colorand flavor ? ETHER, OR OXIDE OF ETHYL.Formula C4H10O, or (C2H5)20, or Et20. Experimental Process.—Into a capacious test-tube put asmall quantity of spirit of wine and about half its bulk of sul-phuric acid, mix and gently warm ; the vapor of ether, recog-nized by its odor, is evolved. Adapt a cork and long bent tubeto the test-tube, and slowly distil over the ether into anothertest-tu Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemistry-general-medical-and-pharmaceutical-including-the-chemistry-of-the-uspharmacopoeia-a-manual-on-the-general-principles-of-the-science-and-their-applications-in-medicine-and-pharmacy-due-its-colorand-flavor-ether-or-oxide-of-ethylformula-c4h10o-or-c2h520-or-et20-experimental-processinto-a-capacious-test-tube-put-asmall-quantity-of-spirit-of-wine-and-about-half-its-bulk-of-sul-phuric-acid-mix-and-gently-warm-the-vapor-of-ether-recog-nized-by-its-odor-is-evolved-adapt-a-cork-and-long-bent-tubeto-the-test-tube-and-slowly-distil-over-the-ether-into-anothertest-tu-image343106156.html
Chemistry : general, medical, and pharmaceutical including the chemistry of the U.Spharmacopoeia ; a manual on the general principles of the science, and their applications in medicine and pharmacy . due its colorand flavor ? ETHER, OR OXIDE OF ETHYL.Formula C4H10O, or (C2H5)20, or Et20. Experimental Process.—Into a capacious test-tube put asmall quantity of spirit of wine and about half its bulk of sul-phuric acid, mix and gently warm ; the vapor of ether, recog-nized by its odor, is evolved. Adapt a cork and long bent tubeto the test-tube, and slowly distil over the ether into anothertest-tu Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemistry-general-medical-and-pharmaceutical-including-the-chemistry-of-the-uspharmacopoeia-a-manual-on-the-general-principles-of-the-science-and-their-applications-in-medicine-and-pharmacy-due-its-colorand-flavor-ether-or-oxide-of-ethylformula-c4h10o-or-c2h520-or-et20-experimental-processinto-a-capacious-test-tube-put-asmall-quantity-of-spirit-of-wine-and-about-half-its-bulk-of-sul-phuric-acid-mix-and-gently-warm-the-vapor-of-ether-recog-nized-by-its-odor-is-evolved-adapt-a-cork-and-long-bent-tubeto-the-test-tube-and-slowly-distil-over-the-ether-into-anothertest-tu-image343106156.htmlRM2AX5RB8–Chemistry : general, medical, and pharmaceutical including the chemistry of the U.Spharmacopoeia ; a manual on the general principles of the science, and their applications in medicine and pharmacy . due its colorand flavor ? ETHER, OR OXIDE OF ETHYL.Formula C4H10O, or (C2H5)20, or Et20. Experimental Process.—Into a capacious test-tube put asmall quantity of spirit of wine and about half its bulk of sul-phuric acid, mix and gently warm ; the vapor of ether, recog-nized by its odor, is evolved. Adapt a cork and long bent tubeto the test-tube, and slowly distil over the ether into anothertest-tu
 Triethylamine A colorless, highly flammable liquid used as a solvent and in the production of various chemicals, such as pharmaceuticals and pesticide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/triethylamine-a-colorless-highly-flammable-liquid-used-as-a-solvent-and-in-the-production-of-various-chemicals-such-as-pharmaceuticals-and-pesticide-image563882928.html
Triethylamine A colorless, highly flammable liquid used as a solvent and in the production of various chemicals, such as pharmaceuticals and pesticide Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/triethylamine-a-colorless-highly-flammable-liquid-used-as-a-solvent-and-in-the-production-of-various-chemicals-such-as-pharmaceuticals-and-pesticide-image563882928.htmlRF2RNB2C0–Triethylamine A colorless, highly flammable liquid used as a solvent and in the production of various chemicals, such as pharmaceuticals and pesticide
 . dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/dan-bai-zhi-fen-zi-ji-chu-botany-2-6-m-so-ltch3coo-ltcrltbr-ltn03-ltc104-lti-ltcns-ch3n-ltnhzltrbknacsltliltmgtquotltca2ltba2-ch3inltc2h5ltnltc3h74nltchgn-sv-ltch3cooltcl-ltbrltc10cns-ch3nnhknaltliltca-ch3nltqh5nltc3hnltch8n-sds-image216141336.html
. dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/dan-bai-zhi-fen-zi-ji-chu-botany-2-6-m-so-ltch3coo-ltcrltbr-ltn03-ltc104-lti-ltcns-ch3n-ltnhzltrbknacsltliltmgtquotltca2ltba2-ch3inltc2h5ltnltc3h74nltchgn-sv-ltch3cooltcl-ltbrltc10cns-ch3nnhknaltliltca-ch3nltqh5nltc3hnltch8n-sds-image216141336.htmlRMPFJ2DC–. dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é
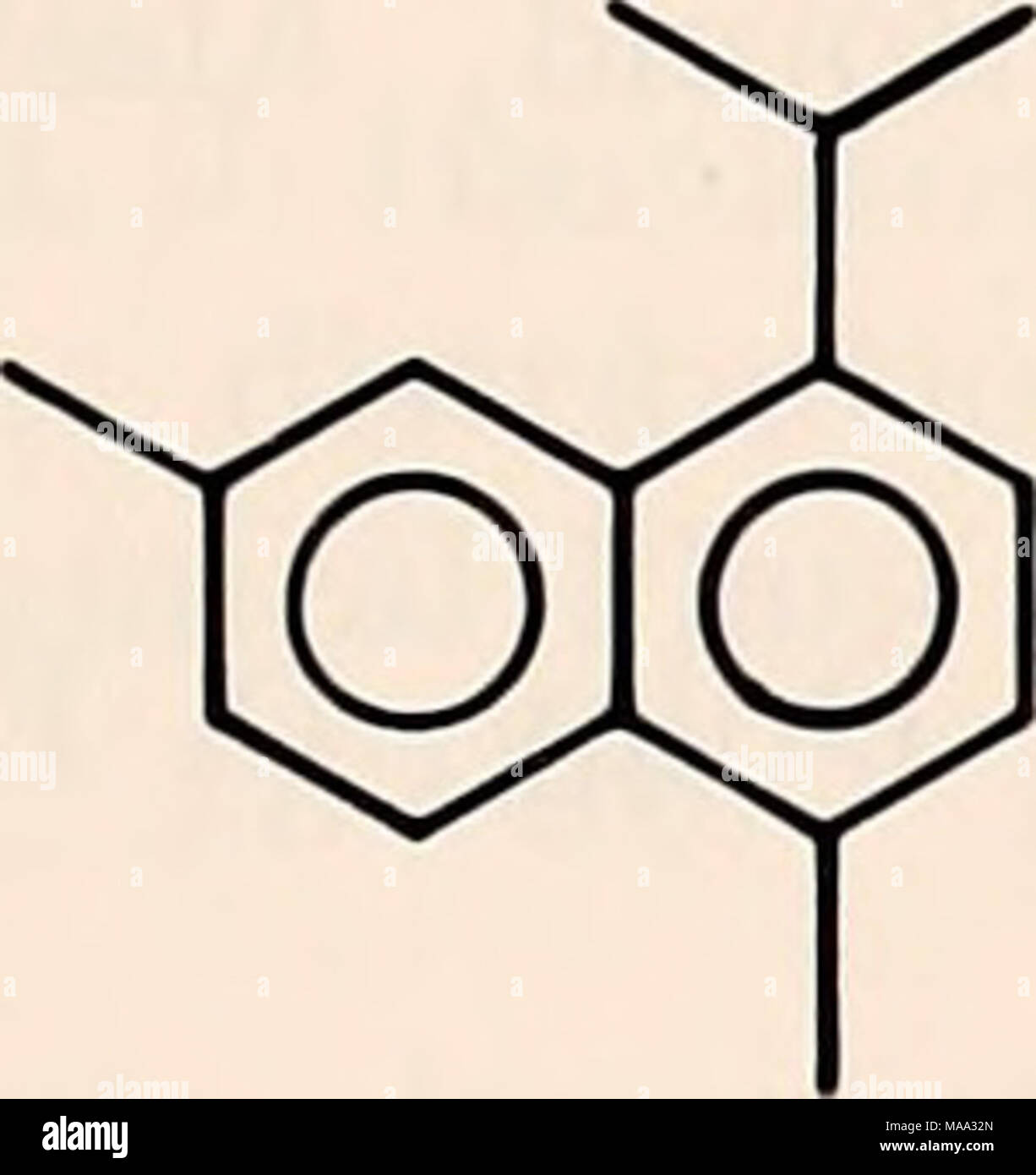 . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-image178472189.html
. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-image178472189.htmlRMMAA32N–. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25
 X-ray . hysical laws of nature. Ponce is often found burning the midnight oil(on Broad Street) and is an adherent of the theorydrink (C2H5 O H) and love beautiful womenfor to-morrow you may die. EDWARD KINLEY ROSEHerald, Va. Eddie Kappa Psi; Pi Theta Sigma; Lin-coln Memorial University; South-west Virginia Club; Pharmaceuti-cal Association. No man is justified in doing efil on ihe groundof expediency. Rose is somewhat of a philosopher. Alwayswilling to do more than his share, and alwayswilling to come to the aid of anyone who needs hishelp, he has made a host of friends since his ar-rival. He Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/x-ray-hysical-laws-of-nature-ponce-is-often-found-burning-the-midnight-oilon-broad-street-and-is-an-adherent-of-the-theorydrink-c2h5-o-h-and-love-beautiful-womenfor-to-morrow-you-may-die-edward-kinley-roseherald-va-eddie-kappa-psi-pi-theta-sigma-lin-coln-memorial-university-south-west-virginia-club-pharmaceuti-cal-association-no-man-is-justified-in-doing-efil-on-ihe-groundof-expediency-rose-is-somewhat-of-a-philosopher-alwayswilling-to-do-more-than-his-share-and-alwayswilling-to-come-to-the-aid-of-anyone-who-needs-hishelp-he-has-made-a-host-of-friends-since-his-ar-rival-he-image339370146.html
X-ray . hysical laws of nature. Ponce is often found burning the midnight oil(on Broad Street) and is an adherent of the theorydrink (C2H5 O H) and love beautiful womenfor to-morrow you may die. EDWARD KINLEY ROSEHerald, Va. Eddie Kappa Psi; Pi Theta Sigma; Lin-coln Memorial University; South-west Virginia Club; Pharmaceuti-cal Association. No man is justified in doing efil on ihe groundof expediency. Rose is somewhat of a philosopher. Alwayswilling to do more than his share, and alwayswilling to come to the aid of anyone who needs hishelp, he has made a host of friends since his ar-rival. He Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/x-ray-hysical-laws-of-nature-ponce-is-often-found-burning-the-midnight-oilon-broad-street-and-is-an-adherent-of-the-theorydrink-c2h5-o-h-and-love-beautiful-womenfor-to-morrow-you-may-die-edward-kinley-roseherald-va-eddie-kappa-psi-pi-theta-sigma-lin-coln-memorial-university-south-west-virginia-club-pharmaceuti-cal-association-no-man-is-justified-in-doing-efil-on-ihe-groundof-expediency-rose-is-somewhat-of-a-philosopher-alwayswilling-to-do-more-than-his-share-and-alwayswilling-to-come-to-the-aid-of-anyone-who-needs-hishelp-he-has-made-a-host-of-friends-since-his-ar-rival-he-image339370146.htmlRM2AM3J2A–X-ray . hysical laws of nature. Ponce is often found burning the midnight oil(on Broad Street) and is an adherent of the theorydrink (C2H5 O H) and love beautiful womenfor to-morrow you may die. EDWARD KINLEY ROSEHerald, Va. Eddie Kappa Psi; Pi Theta Sigma; Lin-coln Memorial University; South-west Virginia Club; Pharmaceuti-cal Association. No man is justified in doing efil on ihe groundof expediency. Rose is somewhat of a philosopher. Alwayswilling to do more than his share, and alwayswilling to come to the aid of anyone who needs hishelp, he has made a host of friends since his ar-rival. He
 . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-rtr-vii-178-h-2l3h-hopanes-r=-h-c2h5-cjhy-viii-extended-17a-h-2l3h-hopanes-r-ch3-c5h11-405-image178472188.html
. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-rtr-vii-178-h-2l3h-hopanes-r=-h-c2h5-cjhy-viii-extended-17a-h-2l3h-hopanes-r-ch3-c5h11-405-image178472188.htmlRMMAA32M–. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder . rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405
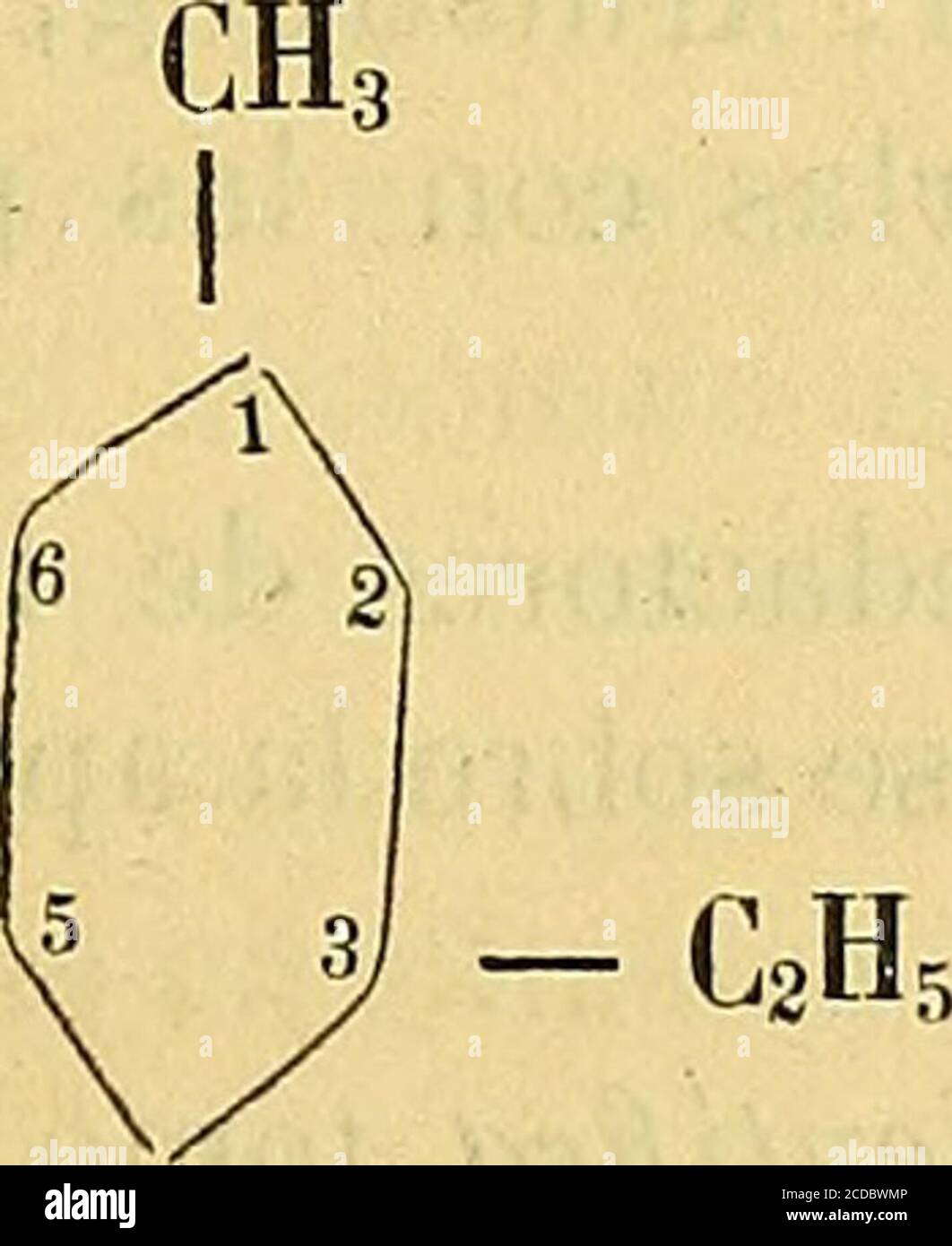 . Anales de la Sociedad Científica Argentina . ^ V — CHoOH A/ 6 2 5 3 - CH2OH 5 3, CO2H. C2H5 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/anales-de-la-sociedad-cientfica-argentina-v-chooh-a-6-2-5-3-ch2oh-5-3-co2h-c2h5-image369669910.html
. Anales de la Sociedad Científica Argentina . ^ V — CHoOH A/ 6 2 5 3 - CH2OH 5 3, CO2H. C2H5 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/anales-de-la-sociedad-cientfica-argentina-v-chooh-a-6-2-5-3-ch2oh-5-3-co2h-c2h5-image369669910.htmlRM2CDBWMP–. Anales de la Sociedad Científica Argentina . ^ V — CHoOH A/ 6 2 5 3 - CH2OH 5 3, CO2H. C2H5
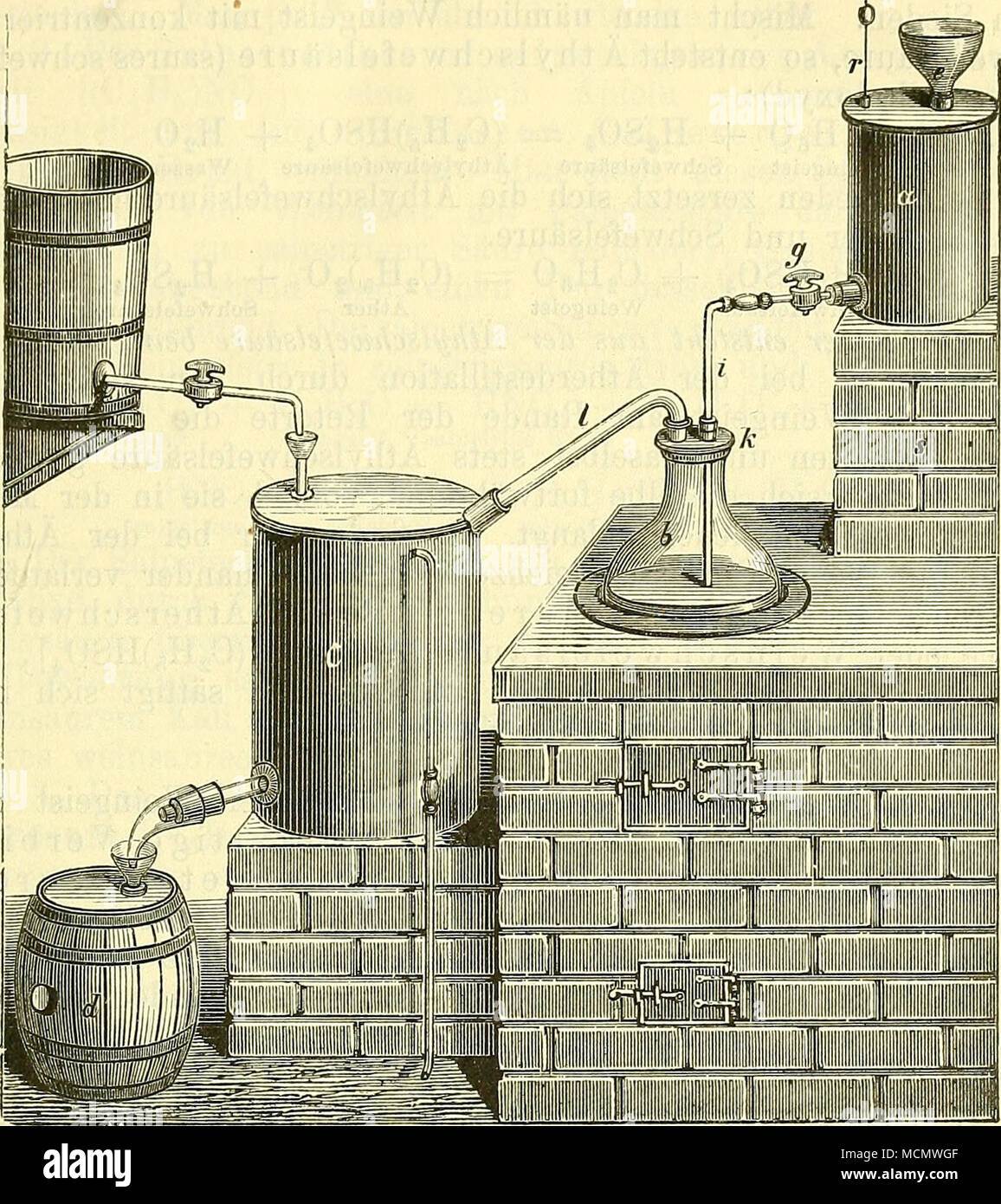 . Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Schwefelsäure in der Wärme geringer als in der Kälte ist, so kann die Säure den Weingeist wohl nicht durch Wasser- entzieh ung ätherifizieren, weil dies nicht in der Kälte ge- schieht. Die Ätherbildung besitzt viel Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/fig-71-theorie-der-therbildung-der-ther-stellt-das-oxyd-des-im-quotweingeist-enthaltenen-thvls-c2h5-dar-c4h1no-=-c2h520-ther-thyloxyd-wie-geht-nun-der-weingeist-thylhydroxyd-in-ther-thyloxyd-ber-frher-glaubte-man-dass-die-konz-schwefel-sure-vermge-ihrer-wasseranziehenden-kraft-dem-weingeist-hydratwasser-entzge-da-aber-die-wasseranziehende-kraft-der-schwefelsure-in-der-wrme-geringer-als-in-der-klte-ist-so-kann-die-sure-den-weingeist-wohl-nicht-durch-wasser-entzieh-ung-therifizieren-weil-dies-nicht-in-der-klte-ge-schieht-die-therbildung-besitzt-viel-image179938655.html
. Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Schwefelsäure in der Wärme geringer als in der Kälte ist, so kann die Säure den Weingeist wohl nicht durch Wasser- entzieh ung ätherifizieren, weil dies nicht in der Kälte ge- schieht. Die Ätherbildung besitzt viel Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/fig-71-theorie-der-therbildung-der-ther-stellt-das-oxyd-des-im-quotweingeist-enthaltenen-thvls-c2h5-dar-c4h1no-=-c2h520-ther-thyloxyd-wie-geht-nun-der-weingeist-thylhydroxyd-in-ther-thyloxyd-ber-frher-glaubte-man-dass-die-konz-schwefel-sure-vermge-ihrer-wasseranziehenden-kraft-dem-weingeist-hydratwasser-entzge-da-aber-die-wasseranziehende-kraft-der-schwefelsure-in-der-wrme-geringer-als-in-der-klte-ist-so-kann-die-sure-den-weingeist-wohl-nicht-durch-wasser-entzieh-ung-therifizieren-weil-dies-nicht-in-der-klte-ge-schieht-die-therbildung-besitzt-viel-image179938655.htmlRMMCMWGF–. Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Schwefelsäure in der Wärme geringer als in der Kälte ist, so kann die Säure den Weingeist wohl nicht durch Wasser- entzieh ung ätherifizieren, weil dies nicht in der Kälte ge- schieht. Die Ätherbildung besitzt viel
 . The biology of marine animals. Marine animals; Physiology, Comparative. OH O. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Nicol, J. A. Colin (Joseph Arthur Colin), 1915-. New York, Interscience Publishers Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biology-of-marine-animals-marine-animals-physiology-comparative-oh-o-pigments-and-colours-oh-o-c2h5-481-oh-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-nicol-j-a-colin-joseph-arthur-colin-1915-new-york-interscience-publishers-image234602399.html
. The biology of marine animals. Marine animals; Physiology, Comparative. OH O. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Nicol, J. A. Colin (Joseph Arthur Colin), 1915-. New York, Interscience Publishers Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biology-of-marine-animals-marine-animals-physiology-comparative-oh-o-pigments-and-colours-oh-o-c2h5-481-oh-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-nicol-j-a-colin-joseph-arthur-colin-1915-new-york-interscience-publishers-image234602399.htmlRMRHK1N3–. The biology of marine animals. Marine animals; Physiology, Comparative. OH O. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Nicol, J. A. Colin (Joseph Arthur Colin), 1915-. New York, Interscience Publishers
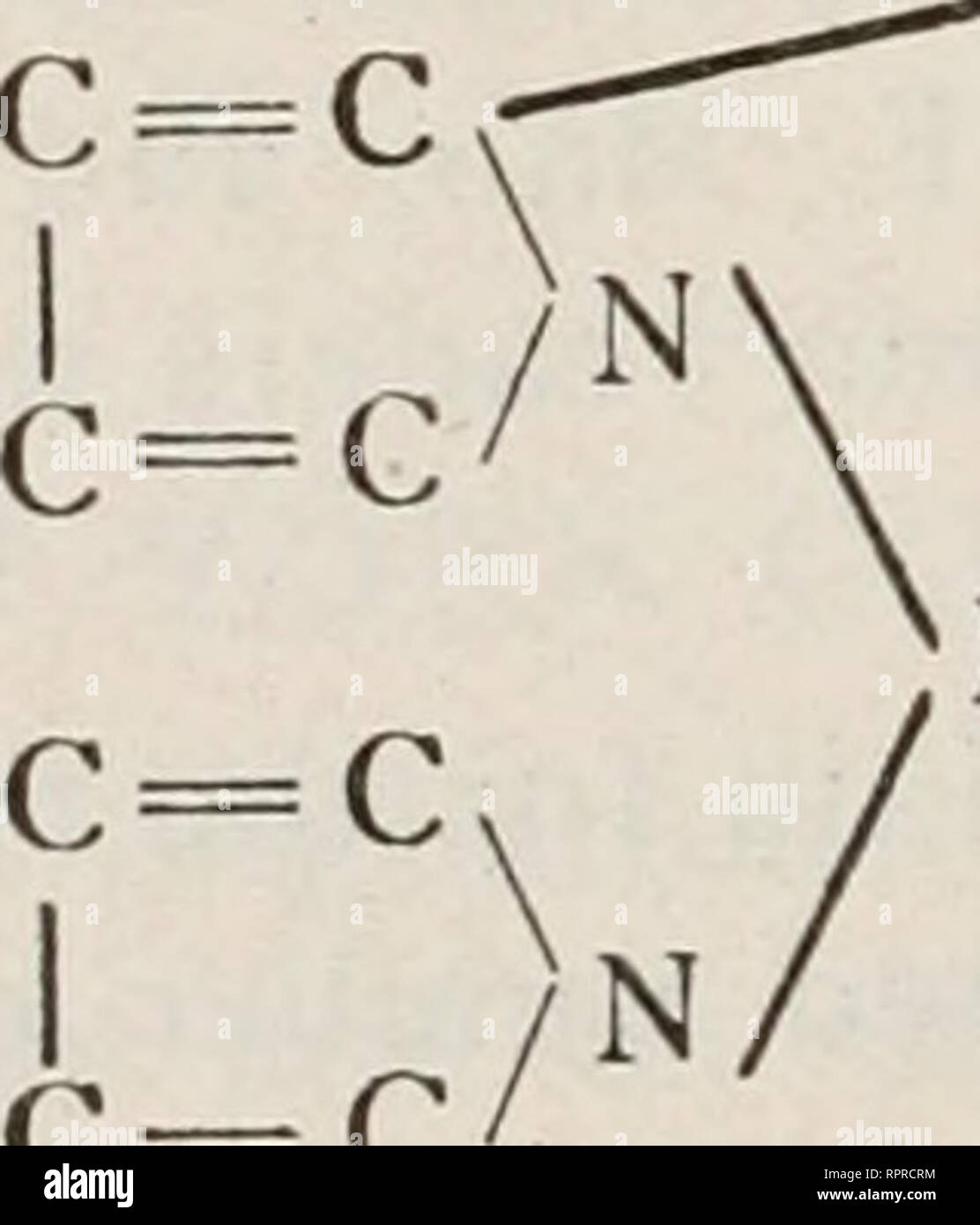 . The Alumni journal. Columbia University. College of Pharmacy; Pharmacology. 74 THE ALUMNI JOURNAL CHLOROPHYLL.* By Curt P. Wimmer, M.A., Phar.D. (Continued.) The question as to whether the chlorophylls of all plants are alike was answered in the affirmative after an examination of several hun- dred different plant extracts. Discrepancies in the results were found to he due to an enzyme action. When a green plant is extracted with alcohol, an enzyme which accompanies the chlorophyll, becomes active and alcoholizes it; the phytol rest is replaced quantitatively by C2H5. The solvent will show a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-alumni-journal-columbia-university-college-of-pharmacy-pharmacology-74-the-alumni-journal-chlorophyll-by-curt-p-wimmer-ma-phard-continued-the-question-as-to-whether-the-chlorophylls-of-all-plants-are-alike-was-answered-in-the-affirmative-after-an-examination-of-several-hun-dred-different-plant-extracts-discrepancies-in-the-results-were-found-to-he-due-to-an-enzyme-action-when-a-green-plant-is-extracted-with-alcohol-an-enzyme-which-accompanies-the-chlorophyll-becomes-active-and-alcoholizes-it-the-phytol-rest-is-replaced-quantitatively-by-c2h5-the-solvent-will-show-a-image237772184.html
. The Alumni journal. Columbia University. College of Pharmacy; Pharmacology. 74 THE ALUMNI JOURNAL CHLOROPHYLL.* By Curt P. Wimmer, M.A., Phar.D. (Continued.) The question as to whether the chlorophylls of all plants are alike was answered in the affirmative after an examination of several hun- dred different plant extracts. Discrepancies in the results were found to he due to an enzyme action. When a green plant is extracted with alcohol, an enzyme which accompanies the chlorophyll, becomes active and alcoholizes it; the phytol rest is replaced quantitatively by C2H5. The solvent will show a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-alumni-journal-columbia-university-college-of-pharmacy-pharmacology-74-the-alumni-journal-chlorophyll-by-curt-p-wimmer-ma-phard-continued-the-question-as-to-whether-the-chlorophylls-of-all-plants-are-alike-was-answered-in-the-affirmative-after-an-examination-of-several-hun-dred-different-plant-extracts-discrepancies-in-the-results-were-found-to-he-due-to-an-enzyme-action-when-a-green-plant-is-extracted-with-alcohol-an-enzyme-which-accompanies-the-chlorophyll-becomes-active-and-alcoholizes-it-the-phytol-rest-is-replaced-quantitatively-by-c2h5-the-solvent-will-show-a-image237772184.htmlRMRPRCRM–. The Alumni journal. Columbia University. College of Pharmacy; Pharmacology. 74 THE ALUMNI JOURNAL CHLOROPHYLL.* By Curt P. Wimmer, M.A., Phar.D. (Continued.) The question as to whether the chlorophylls of all plants are alike was answered in the affirmative after an examination of several hun- dred different plant extracts. Discrepancies in the results were found to he due to an enzyme action. When a green plant is extracted with alcohol, an enzyme which accompanies the chlorophyll, becomes active and alcoholizes it; the phytol rest is replaced quantitatively by C2H5. The solvent will show a
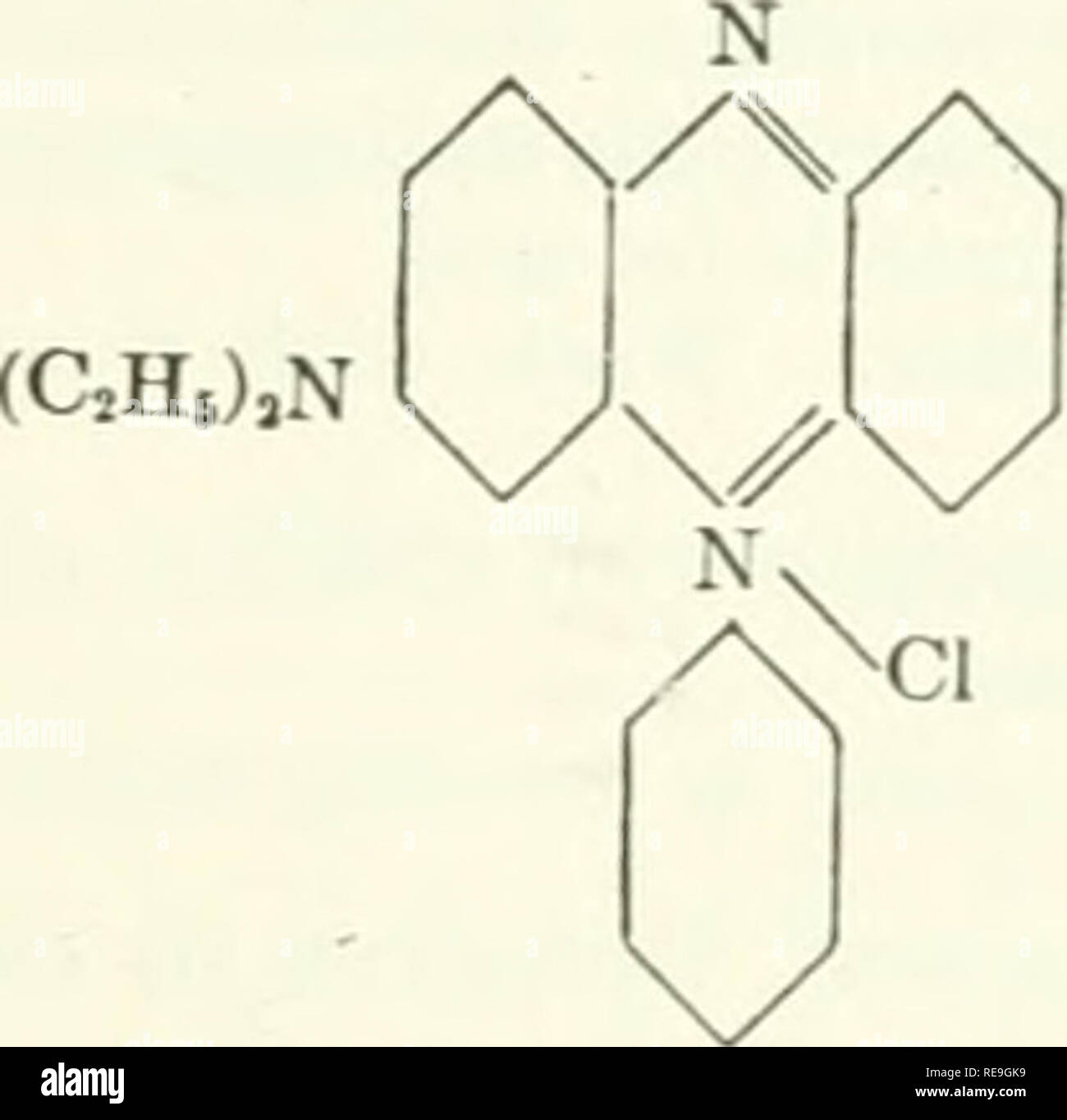
 . Bioenergetics. Bioenergetics. 20 system of conjugated double bonds. One of the major group* a made up of aromatic hydrocarbons, which have no charges and are unable to accept or give off protons. Accordingly, their fluorescence is independent of pH and redox potential. Their absorption lies mostly in the UV. Another important group of fluorescent substances is formed by molecules which have charges, the oscillations of which partake in excitation. The fluorescence of these molecules is sensitive to pH. (C2H5)2N .,,,^^-^0.^^^^ N (C2H5)2CI XOOH Fig. 4. RJiodamin B. and redox potential; their a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bioenergetics-bioenergetics-20-system-of-conjugated-double-bonds-one-of-the-major-group-a-made-up-of-aromatic-hydrocarbons-which-have-no-charges-and-are-unable-to-accept-or-give-off-protons-accordingly-their-fluorescence-is-independent-of-ph-and-redox-potential-their-absorption-lies-mostly-in-the-uv-another-important-group-of-fluorescent-substances-is-formed-by-molecules-which-have-charges-the-oscillations-of-which-partake-in-excitation-the-fluorescence-of-these-molecules-is-sensitive-to-ph-c2h52n-0-n-c2h52ci-xooh-fig-4-rjiodamin-b-and-redox-potential-their-a-image234699318.html
. Bioenergetics. Bioenergetics. 20 system of conjugated double bonds. One of the major group* a made up of aromatic hydrocarbons, which have no charges and are unable to accept or give off protons. Accordingly, their fluorescence is independent of pH and redox potential. Their absorption lies mostly in the UV. Another important group of fluorescent substances is formed by molecules which have charges, the oscillations of which partake in excitation. The fluorescence of these molecules is sensitive to pH. (C2H5)2N .,,,^^-^0.^^^^ N (C2H5)2CI XOOH Fig. 4. RJiodamin B. and redox potential; their a Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/bioenergetics-bioenergetics-20-system-of-conjugated-double-bonds-one-of-the-major-group-a-made-up-of-aromatic-hydrocarbons-which-have-no-charges-and-are-unable-to-accept-or-give-off-protons-accordingly-their-fluorescence-is-independent-of-ph-and-redox-potential-their-absorption-lies-mostly-in-the-uv-another-important-group-of-fluorescent-substances-is-formed-by-molecules-which-have-charges-the-oscillations-of-which-partake-in-excitation-the-fluorescence-of-these-molecules-is-sensitive-to-ph-c2h52n-0-n-c2h52ci-xooh-fig-4-rjiodamin-b-and-redox-potential-their-a-image234699318.htmlRMRHRDAE–. Bioenergetics. Bioenergetics. 20 system of conjugated double bonds. One of the major group* a made up of aromatic hydrocarbons, which have no charges and are unable to accept or give off protons. Accordingly, their fluorescence is independent of pH and redox potential. Their absorption lies mostly in the UV. Another important group of fluorescent substances is formed by molecules which have charges, the oscillations of which partake in excitation. The fluorescence of these molecules is sensitive to pH. (C2H5)2N .,,,^^-^0.^^^^ N (C2H5)2CI XOOH Fig. 4. RJiodamin B. and redox potential; their a
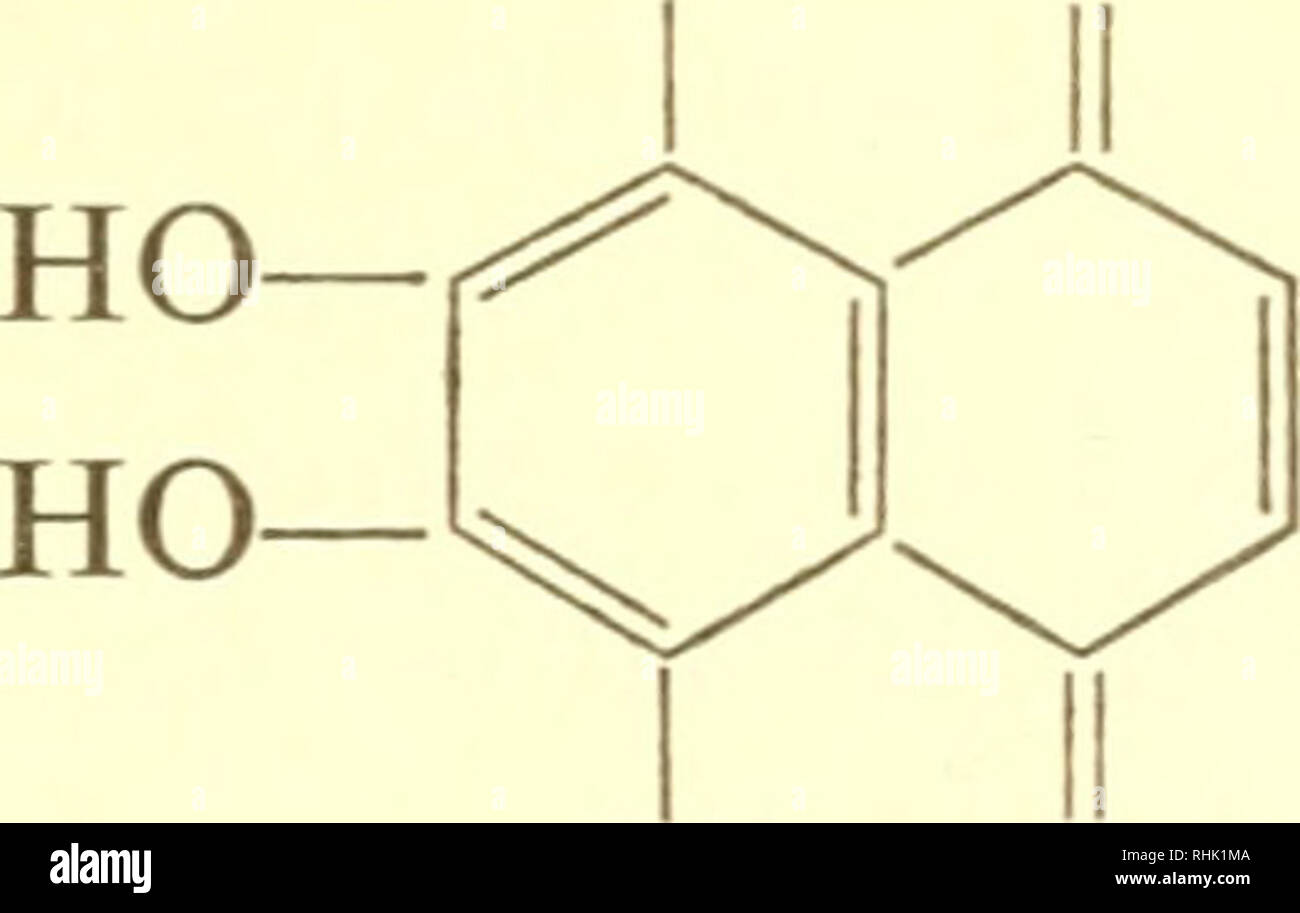 . The biology of marine animals. Marine animals; Physiology, Comparative. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. CHOH—CH3 -OH OH O ECHINOCHROME A OH O SPINOCHROME A Indoles. These include indigoid pigments and melanins, basically sub- stances containing a phenopyrrol nucleus. Indigoid pigments are produced by certain gastropods, and include indigo and the purple dye dibromindigo, secreted by Murex, Mitra and Nucella. This substance is the dyestuff Tyrian purple which was used so extensively by the ancients. It is produced by a special hypobranchial gland, and stored as the colourless leuco c Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biology-of-marine-animals-marine-animals-physiology-comparative-pigments-and-colours-oh-o-c2h5-481-oh-chohch3-oh-oh-o-echinochrome-a-oh-o-spinochrome-a-indoles-these-include-indigoid-pigments-and-melanins-basically-sub-stances-containing-a-phenopyrrol-nucleus-indigoid-pigments-are-produced-by-certain-gastropods-and-include-indigo-and-the-purple-dye-dibromindigo-secreted-by-murex-mitra-and-nucella-this-substance-is-the-dyestuff-tyrian-purple-which-was-used-so-extensively-by-the-ancients-it-is-produced-by-a-special-hypobranchial-gland-and-stored-as-the-colourless-leuco-c-image234602378.html
. The biology of marine animals. Marine animals; Physiology, Comparative. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. CHOH—CH3 -OH OH O ECHINOCHROME A OH O SPINOCHROME A Indoles. These include indigoid pigments and melanins, basically sub- stances containing a phenopyrrol nucleus. Indigoid pigments are produced by certain gastropods, and include indigo and the purple dye dibromindigo, secreted by Murex, Mitra and Nucella. This substance is the dyestuff Tyrian purple which was used so extensively by the ancients. It is produced by a special hypobranchial gland, and stored as the colourless leuco c Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biology-of-marine-animals-marine-animals-physiology-comparative-pigments-and-colours-oh-o-c2h5-481-oh-chohch3-oh-oh-o-echinochrome-a-oh-o-spinochrome-a-indoles-these-include-indigoid-pigments-and-melanins-basically-sub-stances-containing-a-phenopyrrol-nucleus-indigoid-pigments-are-produced-by-certain-gastropods-and-include-indigo-and-the-purple-dye-dibromindigo-secreted-by-murex-mitra-and-nucella-this-substance-is-the-dyestuff-tyrian-purple-which-was-used-so-extensively-by-the-ancients-it-is-produced-by-a-special-hypobranchial-gland-and-stored-as-the-colourless-leuco-c-image234602378.htmlRMRHK1MA–. The biology of marine animals. Marine animals; Physiology, Comparative. PIGMENTS AND COLOURS OH O —C2H5 481 —OH. CHOH—CH3 -OH OH O ECHINOCHROME A OH O SPINOCHROME A Indoles. These include indigoid pigments and melanins, basically sub- stances containing a phenopyrrol nucleus. Indigoid pigments are produced by certain gastropods, and include indigo and the purple dye dibromindigo, secreted by Murex, Mitra and Nucella. This substance is the dyestuff Tyrian purple which was used so extensively by the ancients. It is produced by a special hypobranchial gland, and stored as the colourless leuco c
 . Biochemical systematics. Biochemical variation; Botany. Current Revisions for BIOCHEmCAL SYSTEMATICS p. 110 Lath5rrine heterocyclic ring is aromatic. p. 158 Anhalonidin N-containing ring is saturated, p. 159 Berberine cation should appear as follows:. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearan Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemical-systematics-biochemical-variation-botany-current-revisions-for-biochemcal-systematics-p-110-lath5rrine-heterocyclic-ring-is-aromatic-p-158-anhalonidin-n-containing-ring-is-saturated-p-159-berberine-cation-should-appear-as-follows-p-161-lstonine-skeleton-hydrolysis-product-of-physostigmine-for-rauwolfine-substitute-established-ajmaline-structure-n-oh-cho-p-162-p-257-gentianin-should-appear-as-follows-c2h5-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearan-image234700147.html
. Biochemical systematics. Biochemical variation; Botany. Current Revisions for BIOCHEmCAL SYSTEMATICS p. 110 Lath5rrine heterocyclic ring is aromatic. p. 158 Anhalonidin N-containing ring is saturated, p. 159 Berberine cation should appear as follows:. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearan Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemical-systematics-biochemical-variation-botany-current-revisions-for-biochemcal-systematics-p-110-lath5rrine-heterocyclic-ring-is-aromatic-p-158-anhalonidin-n-containing-ring-is-saturated-p-159-berberine-cation-should-appear-as-follows-p-161-lstonine-skeleton-hydrolysis-product-of-physostigmine-for-rauwolfine-substitute-established-ajmaline-structure-n-oh-cho-p-162-p-257-gentianin-should-appear-as-follows-c2h5-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearan-image234700147.htmlRMRHREC3–. Biochemical systematics. Biochemical variation; Botany. Current Revisions for BIOCHEmCAL SYSTEMATICS p. 110 Lath5rrine heterocyclic ring is aromatic. p. 158 Anhalonidin N-containing ring is saturated, p. 159 Berberine cation should appear as follows:. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearan
 . Contributions to embryology. Embryology. Fig. F.. (3) Janus green B (Farbwerke Hoechst Company), diethylsafraninazodi- methylanilinchloride: ^' = ^ f ^ N (CH,), Fio. G. The specificity of the reaction is shown by the fact that only the latter, janus green B. the one originally recommended by Michaelis, will stain mitochondria, though the others differ only in the substitution of an Hi or (CH3)2 in the place of the (C2H5)2 group. The presence of the diethyl group in the safranin molecule is e^'idently the determining factor. Compounds containing two ethyl groups are more basic than those cont Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/contributions-to-embryology-embryology-fig-f-3-janus-green-b-farbwerke-hoechst-company-diethylsafraninazodi-methylanilinchloride-=-f-n-ch-fio-g-the-specificity-of-the-reaction-is-shown-by-the-fact-that-only-the-latter-janus-green-b-the-one-originally-recommended-by-michaelis-will-stain-mitochondria-though-the-others-differ-only-in-the-substitution-of-an-hi-or-ch32-in-the-place-of-the-c2h52-group-the-presence-of-the-diethyl-group-in-the-safranin-molecule-is-eidently-the-determining-factor-compounds-containing-two-ethyl-groups-are-more-basic-than-those-cont-image232550621.html
. Contributions to embryology. Embryology. Fig. F.. (3) Janus green B (Farbwerke Hoechst Company), diethylsafraninazodi- methylanilinchloride: ^' = ^ f ^ N (CH,), Fio. G. The specificity of the reaction is shown by the fact that only the latter, janus green B. the one originally recommended by Michaelis, will stain mitochondria, though the others differ only in the substitution of an Hi or (CH3)2 in the place of the (C2H5)2 group. The presence of the diethyl group in the safranin molecule is e^'idently the determining factor. Compounds containing two ethyl groups are more basic than those cont Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/contributions-to-embryology-embryology-fig-f-3-janus-green-b-farbwerke-hoechst-company-diethylsafraninazodi-methylanilinchloride-=-f-n-ch-fio-g-the-specificity-of-the-reaction-is-shown-by-the-fact-that-only-the-latter-janus-green-b-the-one-originally-recommended-by-michaelis-will-stain-mitochondria-though-the-others-differ-only-in-the-substitution-of-an-hi-or-ch32-in-the-place-of-the-c2h52-group-the-presence-of-the-diethyl-group-in-the-safranin-molecule-is-eidently-the-determining-factor-compounds-containing-two-ethyl-groups-are-more-basic-than-those-cont-image232550621.htmlRMRE9GK9–. Contributions to embryology. Embryology. Fig. F.. (3) Janus green B (Farbwerke Hoechst Company), diethylsafraninazodi- methylanilinchloride: ^' = ^ f ^ N (CH,), Fio. G. The specificity of the reaction is shown by the fact that only the latter, janus green B. the one originally recommended by Michaelis, will stain mitochondria, though the others differ only in the substitution of an Hi or (CH3)2 in the place of the (C2H5)2 group. The presence of the diethyl group in the safranin molecule is e^'idently the determining factor. Compounds containing two ethyl groups are more basic than those cont
 . The Biological bulletin. Biology; Zoology; Biology; Marine Biology. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. COOCHo COOCH- FIGURE 1. List of the compounds used for assays of juvenile hormone activity. and Dermestes the compounds were injected in a 1 (A drop of olive oil into the body cavity of freshly molted pupae. The activity was determined according to the degree of retention of the larval (Hemiptera) or pupal (Coleoptera) characters after the next ecdysis. The dose-response experiments performed on Pyrrhocoris and Graphosoma revealed that the whole range of activity from zero Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-coor-cooch-cooch-xii-r-=-ch3-xiii-r-=-c2h5-xiv-xv-coocho-cooch-figure-1-list-of-the-compounds-used-for-assays-of-juvenile-hormone-activity-and-dermestes-the-compounds-were-injected-in-a-1-a-drop-of-olive-oil-into-the-body-cavity-of-freshly-molted-pupae-the-activity-was-determined-according-to-the-degree-of-retention-of-the-larval-hemiptera-or-pupal-coleoptera-characters-after-the-next-ecdysis-the-dose-response-experiments-performed-on-pyrrhocoris-and-graphosoma-revealed-that-the-whole-range-of-activity-from-zero-image234651746.html
. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. COOCHo COOCH- FIGURE 1. List of the compounds used for assays of juvenile hormone activity. and Dermestes the compounds were injected in a 1 (A drop of olive oil into the body cavity of freshly molted pupae. The activity was determined according to the degree of retention of the larval (Hemiptera) or pupal (Coleoptera) characters after the next ecdysis. The dose-response experiments performed on Pyrrhocoris and Graphosoma revealed that the whole range of activity from zero Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-coor-cooch-cooch-xii-r-=-ch3-xiii-r-=-c2h5-xiv-xv-coocho-cooch-figure-1-list-of-the-compounds-used-for-assays-of-juvenile-hormone-activity-and-dermestes-the-compounds-were-injected-in-a-1-a-drop-of-olive-oil-into-the-body-cavity-of-freshly-molted-pupae-the-activity-was-determined-according-to-the-degree-of-retention-of-the-larval-hemiptera-or-pupal-coleoptera-characters-after-the-next-ecdysis-the-dose-response-experiments-performed-on-pyrrhocoris-and-graphosoma-revealed-that-the-whole-range-of-activity-from-zero-image234651746.htmlRMRHN8KE–. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. COOCHo COOCH- FIGURE 1. List of the compounds used for assays of juvenile hormone activity. and Dermestes the compounds were injected in a 1 (A drop of olive oil into the body cavity of freshly molted pupae. The activity was determined according to the degree of retention of the larval (Hemiptera) or pupal (Coleoptera) characters after the next ecdysis. The dose-response experiments performed on Pyrrhocoris and Graphosoma revealed that the whole range of activity from zero
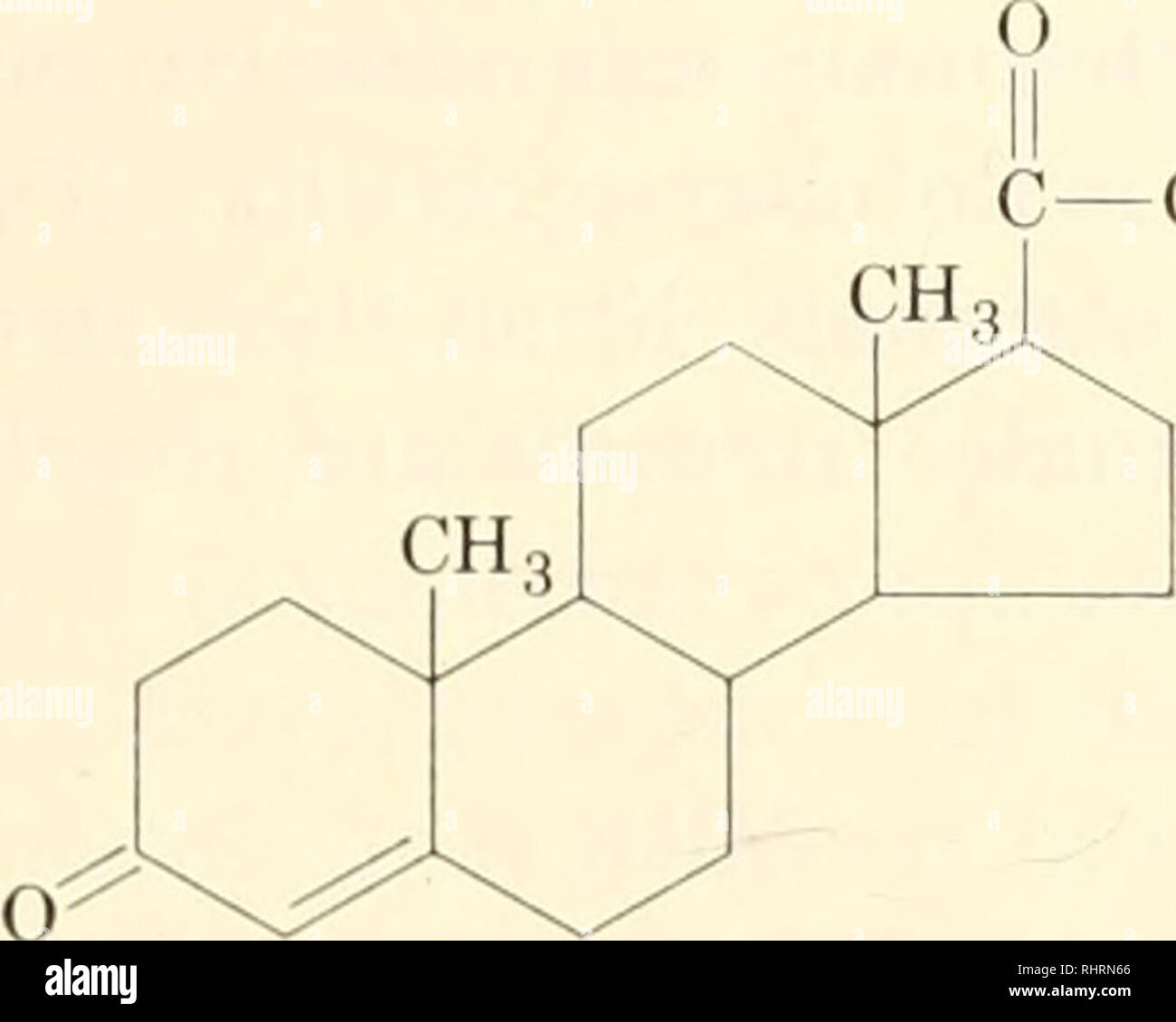 . Biochemistry of plants and animals, an introduction. Biochemistry. BODY TISSUES 323. CH., progesterone Stilbestrol is a synthetic estrogen derived from stilbene (C^Hj-CH : CH-CcHr,). It is not chemically related to the natural estrogens, but the diethyl derivative is about three times as potent as estrone. This compound has found wide use in modern medical practice as a supple- ment to the natural supply of estrogens. C,H5 HO- -C=C-/ -OH C2H5 dietliylslilbestrol Many plants contain estrogenic substances in varying quantities which may be factors in the growth of animals. The administration Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemistry-of-plants-and-animals-an-introduction-biochemistry-body-tissues-323-ch-progesterone-stilbestrol-is-a-synthetic-estrogen-derived-from-stilbene-chj-ch-ch-cchr-it-is-not-chemically-related-to-the-natural-estrogens-but-the-diethyl-derivative-is-about-three-times-as-potent-as-estrone-this-compound-has-found-wide-use-in-modern-medical-practice-as-a-supple-ment-to-the-natural-supply-of-estrogens-ch5-ho-c=c-oh-c2h5-dietliylslilbestrol-many-plants-contain-estrogenic-substances-in-varying-quantities-which-may-be-factors-in-the-growth-of-animals-the-administration-image234705470.html
. Biochemistry of plants and animals, an introduction. Biochemistry. BODY TISSUES 323. CH., progesterone Stilbestrol is a synthetic estrogen derived from stilbene (C^Hj-CH : CH-CcHr,). It is not chemically related to the natural estrogens, but the diethyl derivative is about three times as potent as estrone. This compound has found wide use in modern medical practice as a supple- ment to the natural supply of estrogens. C,H5 HO- -C=C-/ -OH C2H5 dietliylslilbestrol Many plants contain estrogenic substances in varying quantities which may be factors in the growth of animals. The administration Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemistry-of-plants-and-animals-an-introduction-biochemistry-body-tissues-323-ch-progesterone-stilbestrol-is-a-synthetic-estrogen-derived-from-stilbene-chj-ch-ch-cchr-it-is-not-chemically-related-to-the-natural-estrogens-but-the-diethyl-derivative-is-about-three-times-as-potent-as-estrone-this-compound-has-found-wide-use-in-modern-medical-practice-as-a-supple-ment-to-the-natural-supply-of-estrogens-ch5-ho-c=c-oh-c2h5-dietliylslilbestrol-many-plants-contain-estrogenic-substances-in-varying-quantities-which-may-be-factors-in-the-growth-of-animals-the-administration-image234705470.htmlRMRHRN66–. Biochemistry of plants and animals, an introduction. Biochemistry. BODY TISSUES 323. CH., progesterone Stilbestrol is a synthetic estrogen derived from stilbene (C^Hj-CH : CH-CcHr,). It is not chemically related to the natural estrogens, but the diethyl derivative is about three times as potent as estrone. This compound has found wide use in modern medical practice as a supple- ment to the natural supply of estrogens. C,H5 HO- -C=C-/ -OH C2H5 dietliylslilbestrol Many plants contain estrogenic substances in varying quantities which may be factors in the growth of animals. The administration
 . The Canadian record of science. Natural history. 304 Canadian Record of Science. According to the researches of Froelich,* the methyl groups in pseudo-cumidine, occupy the positions 2, 4 and 5, the amido group being 1. There are thus only the positions 3 and 6 left for the ethyl groups to fill. The constitutional formula for the base is therefore— NIL C2H5. CEL On the occurrence of Scolithus in Rocks of the Ohazy Formation about Ottawa, Ontario. By Henry M. Ami, M.A., F.G.S. For years past, the occurrence of Scolithus in the lower portion of the Cambro-Silurian or Ordovician strata, as it is Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-canadian-record-of-science-natural-history-304-canadian-record-of-science-according-to-the-researches-of-froelich-the-methyl-groups-in-pseudo-cumidine-occupy-the-positions-2-4-and-5-the-amido-group-being-1-there-are-thus-only-the-positions-3-and-6-left-for-the-ethyl-groups-to-fill-the-constitutional-formula-for-the-base-is-therefore-nil-c2h5-cel-on-the-occurrence-of-scolithus-in-rocks-of-the-ohazy-formation-about-ottawa-ontario-by-henry-m-ami-ma-fgs-for-years-past-the-occurrence-of-scolithus-in-the-lower-portion-of-the-cambro-silurian-or-ordovician-strata-as-it-is-image233531807.html
. The Canadian record of science. Natural history. 304 Canadian Record of Science. According to the researches of Froelich,* the methyl groups in pseudo-cumidine, occupy the positions 2, 4 and 5, the amido group being 1. There are thus only the positions 3 and 6 left for the ethyl groups to fill. The constitutional formula for the base is therefore— NIL C2H5. CEL On the occurrence of Scolithus in Rocks of the Ohazy Formation about Ottawa, Ontario. By Henry M. Ami, M.A., F.G.S. For years past, the occurrence of Scolithus in the lower portion of the Cambro-Silurian or Ordovician strata, as it is Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-canadian-record-of-science-natural-history-304-canadian-record-of-science-according-to-the-researches-of-froelich-the-methyl-groups-in-pseudo-cumidine-occupy-the-positions-2-4-and-5-the-amido-group-being-1-there-are-thus-only-the-positions-3-and-6-left-for-the-ethyl-groups-to-fill-the-constitutional-formula-for-the-base-is-therefore-nil-c2h5-cel-on-the-occurrence-of-scolithus-in-rocks-of-the-ohazy-formation-about-ottawa-ontario-by-henry-m-ami-ma-fgs-for-years-past-the-occurrence-of-scolithus-in-the-lower-portion-of-the-cambro-silurian-or-ordovician-strata-as-it-is-image233531807.htmlRMRFX85K–. The Canadian record of science. Natural history. 304 Canadian Record of Science. According to the researches of Froelich,* the methyl groups in pseudo-cumidine, occupy the positions 2, 4 and 5, the amido group being 1. There are thus only the positions 3 and 6 left for the ethyl groups to fill. The constitutional formula for the base is therefore— NIL C2H5. CEL On the occurrence of Scolithus in Rocks of the Ohazy Formation about Ottawa, Ontario. By Henry M. Ami, M.A., F.G.S. For years past, the occurrence of Scolithus in the lower portion of the Cambro-Silurian or Ordovician strata, as it is
![. The Biological bulletin. Biology; Zoology; Marine biology. CH2OH. /// R=C2H5 IV R= benzyl. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photo . The Biological bulletin. Biology; Zoology; Marine biology. CH2OH. /// R=C2H5 IV R= benzyl. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photo](https://c8.alamy.com/comp/RHM43H/the-biological-bulletin-biology-zoology-marine-biology-ch2oh-r=c2h5-iv-r=-benzyl-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-RHM43H.jpg) . The Biological bulletin. Biology; Zoology; Marine biology. CH2OH. /// R=C2H5 IV R= benzyl. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-marine-biology-ch2oh-r=c2h5-iv-r=-benzyl-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-image234626213.html
. The Biological bulletin. Biology; Zoology; Marine biology. CH2OH. /// R=C2H5 IV R= benzyl. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-marine-biology-ch2oh-r=c2h5-iv-r=-benzyl-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-image234626213.htmlRMRHM43H–. The Biological bulletin. Biology; Zoology; Marine biology. CH2OH. /// R=C2H5 IV R= benzyl. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ]
![. The Biological bulletin. Biology; Zoology; Marine biology. /// R=C2H5 IV R= benzyl. COOCH.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photo . The Biological bulletin. Biology; Zoology; Marine biology. /// R=C2H5 IV R= benzyl. COOCH.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photo](https://c8.alamy.com/comp/RHG771/the-biological-bulletin-biology-zoology-marine-biology-r=c2h5-iv-r=-benzyl-cooch-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-RHG771.jpg) . The Biological bulletin. Biology; Zoology; Marine biology. /// R=C2H5 IV R= benzyl. COOCH.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-marine-biology-r=c2h5-iv-r=-benzyl-cooch-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-image234540853.html
. The Biological bulletin. Biology; Zoology; Marine biology. /// R=C2H5 IV R= benzyl. COOCH.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ] Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-marine-biology-r=c2h5-iv-r=-benzyl-cooch-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-clarence-1890-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-highwire-press-lancaster-pa-etc-lancaster-press-inc-etc-image234540853.htmlRMRHG771–. The Biological bulletin. Biology; Zoology; Marine biology. /// R=C2H5 IV R= benzyl. COOCH.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Clarence, 1890-; Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report; HighWire Press. Lancaster, Pa. [etc. ] : Lancaster Press, inc. [etc. ]
 . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. I. retene, CiqHiq II. simonellite, C19H24. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Manag Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-i-retene-ciqhiq-ii-simonellite-c19h24-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-manag-image232458107.html
. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. I. retene, CiqHiq II. simonellite, C19H24. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Manag Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-i-retene-ciqhiq-ii-simonellite-c19h24-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-manag-image232458107.htmlRMRE5AK7–. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. I. retene, CiqHiq II. simonellite, C19H24. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Manag
![. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. <. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Management. [Rockville, Md. ?] : U. S. Dep Stock Photo . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. <. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Management. [Rockville, Md. ?] : U. S. Dep Stock Photo](https://c8.alamy.com/comp/RE5AK6/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-lt-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-management-rockville-md-u-s-dep-RE5AK6.jpg) . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. <. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Management. [Rockville, Md. ?] : U. S. Dep Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-lt-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-management-rockville-md-u-s-dep-image232458106.html
. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. <. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Management. [Rockville, Md. ?] : U. S. Dep Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-iii-cadalene-c15h-18-iv-extended-diterpanes-r=c2h5-c2h25-lt-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-management-rockville-md-u-s-dep-image232458106.htmlRMRE5AK6–. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. III. cadalene, C15H 18 IV. extended diterpanes R=C2H5-C|2H25. <. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Management. [Rockville, Md. ?] : U. S. Dep
 . The Biological bulletin. Biology; Zoology; Biology; Marine Biology. JUVENILE HORMONE ACTIVITY 111 COOCH- IV V R = R = R = R = C2H5 CH3 CoHc I//. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-juvenile-hormone-activity-111-cooch-iv-v-r-=-r-=-r-=-r-=-c2h5-ch3-cohc-i-coor-coor-cooch-cooch-viii-ix-r-=-r-c2h5-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-190708-1952-lillie-frank-rattray-1870-1947-moore-carl-richard-image234651777.html
. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. JUVENILE HORMONE ACTIVITY 111 COOCH- IV V R = R = R = R = C2H5 CH3 CoHc I//. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-juvenile-hormone-activity-111-cooch-iv-v-r-=-r-=-r-=-r-=-c2h5-ch3-cohc-i-coor-coor-cooch-cooch-viii-ix-r-=-r-c2h5-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-190708-1952-lillie-frank-rattray-1870-1947-moore-carl-richard-image234651777.htmlRMRHN8MH–. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. JUVENILE HORMONE ACTIVITY 111 COOCH- IV V R = R = R = R = C2H5 CH3 CoHc I//. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard,
 . The Biological bulletin. Biology; Zoology; Biology; Marine Biology. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-coor-coor-cooch-cooch-viii-ix-r-=-r-c2h5-coor-cooch-cooch-xii-r-=-ch3-xiii-r-=-c2h5-xiv-xv-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-190708-1952-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-image234651759.html
. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-biological-bulletin-biology-zoology-biology-marine-biology-coor-coor-cooch-cooch-viii-ix-r-=-r-c2h5-coor-cooch-cooch-xii-r-=-ch3-xiii-r-=-c2h5-xiv-xv-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-marine-biological-laboratory-woods-hole-mass-marine-biological-laboratory-woods-hole-mass-annual-report-190708-1952-lillie-frank-rattray-1870-1947-moore-carl-richard-1892-redfield-alfred-image234651759.htmlRMRHN8KY–. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. • COOR COOR COOCH, COOCH: VIII IX R = R - C2H5. COOR COOCH- COOCH. XII R = CH3 XIII R = C2H5 XIV XV. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Marine Biological Laboratory (Woods Hole, Mass. ); Marine Biological Laboratory (Woods Hole, Mass. ). Annual report 1907/08-1952; Lillie, Frank Rattray, 1870-1947; Moore, Carl Richard, 1892-; Redfield, Alfred
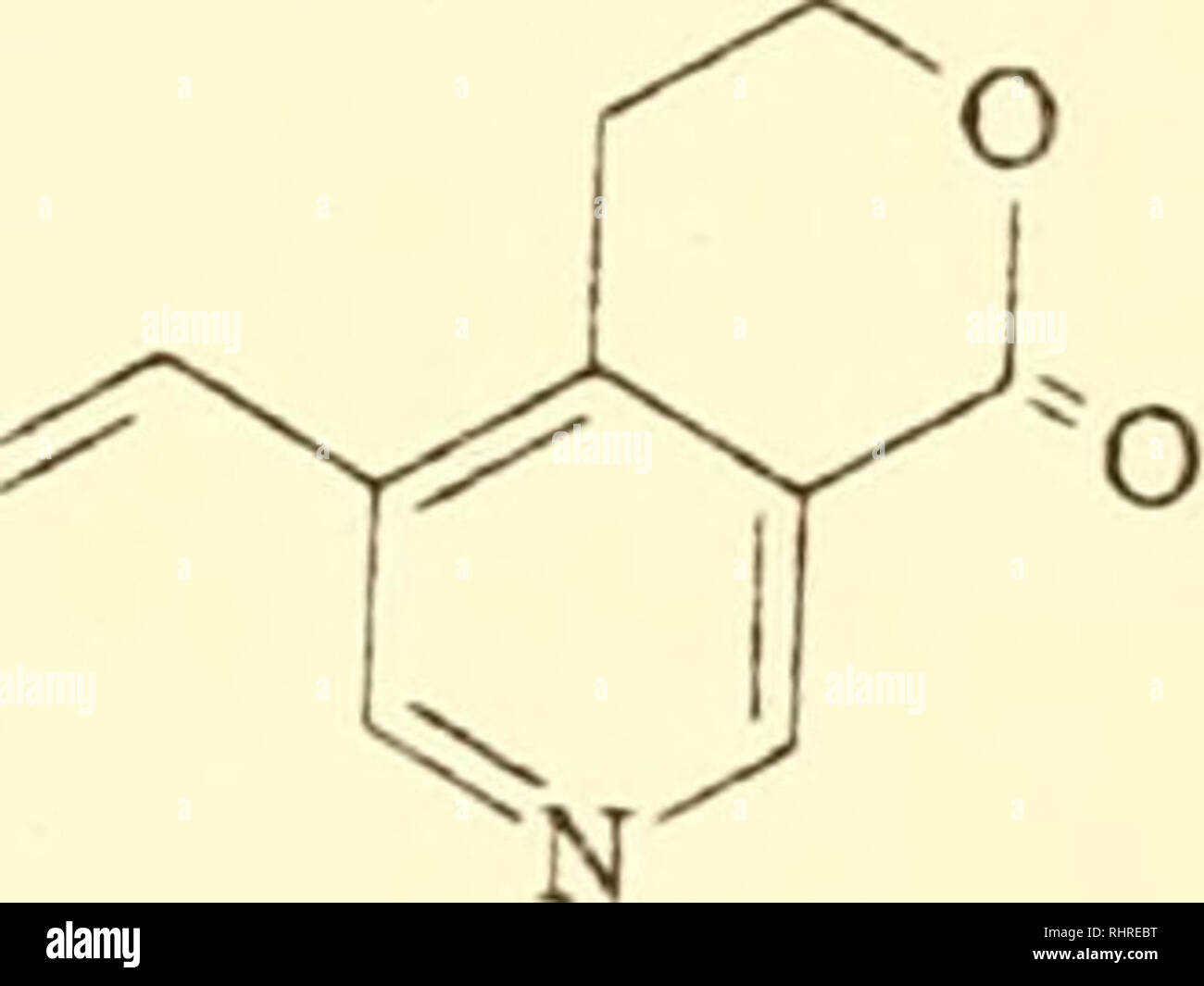 . Biochemical systematics. Biochemical variation; Botany. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. p. 163 Aconitine contains a C^ methoxyl group. p. 176 Ceveratrum and jerveratrum skeletons contain OH at Ci-,- CHo °touds at ^10- *^13' ^20 andC25 p. 197 Rotenone C2-C3 bond is saturated, p. 200 Apigenin lacks 3' -OH. p. 216 Structure 2 has 3-OH. 5-OCH3 substituents; pinostrobin has 7-OCH. structure 5 should read flavanonols. "*' p. 217 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemical-systematics-biochemical-variation-botany-p-161-lstonine-skeleton-hydrolysis-product-of-physostigmine-for-rauwolfine-substitute-established-ajmaline-structure-n-oh-cho-p-162-p-257-gentianin-should-appear-as-follows-c2h5-p-163-aconitine-contains-a-c-methoxyl-group-p-176-ceveratrum-and-jerveratrum-skeletons-contain-oh-at-ci-cho-touds-at-10-13-20-andc25-p-197-rotenone-c2-c3-bond-is-saturated-p-200-apigenin-lacks-3-oh-p-216-structure-2-has-3-oh-5-och3-substituents-pinostrobin-has-7-och-structure-5-should-read-flavanonols-quot-p-217-image234700140.html
. Biochemical systematics. Biochemical variation; Botany. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. p. 163 Aconitine contains a C^ methoxyl group. p. 176 Ceveratrum and jerveratrum skeletons contain OH at Ci-,- CHo °touds at ^10- *^13' ^20 andC25 p. 197 Rotenone C2-C3 bond is saturated, p. 200 Apigenin lacks 3' -OH. p. 216 Structure 2 has 3-OH. 5-OCH3 substituents; pinostrobin has 7-OCH. structure 5 should read flavanonols. "*' p. 217 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemical-systematics-biochemical-variation-botany-p-161-lstonine-skeleton-hydrolysis-product-of-physostigmine-for-rauwolfine-substitute-established-ajmaline-structure-n-oh-cho-p-162-p-257-gentianin-should-appear-as-follows-c2h5-p-163-aconitine-contains-a-c-methoxyl-group-p-176-ceveratrum-and-jerveratrum-skeletons-contain-oh-at-ci-cho-touds-at-10-13-20-andc25-p-197-rotenone-c2-c3-bond-is-saturated-p-200-apigenin-lacks-3-oh-p-216-structure-2-has-3-oh-5-och3-substituents-pinostrobin-has-7-och-structure-5-should-read-flavanonols-quot-p-217-image234700140.htmlRMRHREBT–. Biochemical systematics. Biochemical variation; Botany. p. 161 .lstonine skeleton; hydrolysis product of physostigmine; for rauwolfine substitute established ajmaline structure; N/ -^^^^ OH CHo p. 162, p. 257 Gentianin should appear as follows: ^^C2H5. p. 163 Aconitine contains a C^ methoxyl group. p. 176 Ceveratrum and jerveratrum skeletons contain OH at Ci-,- CHo °touds at ^10- *^13' ^20 andC25 p. 197 Rotenone C2-C3 bond is saturated, p. 200 Apigenin lacks 3' -OH. p. 216 Structure 2 has 3-OH. 5-OCH3 substituents; pinostrobin has 7-OCH. structure 5 should read flavanonols. "*' p. 217
 . The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-rtr-vii-178-h-2l3h-hopanes-r=-h-c2h5-cjhy-viii-extended-17a-h-2l3h-hopanes-r-ch3-c5h11-405-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-image232458103.html
. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/the-eastern-bering-sea-shelf-oceanography-and-resources-edited-by-donald-w-hood-and-john-a-calder-oceanography-bering-sea-rtr-vii-178-h-2l3h-hopanes-r=-h-c2h5-cjhy-viii-extended-17a-h-2l3h-hopanes-r-ch3-c5h11-405-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-hood-d-w-donald-wilbur-1918-calder-john-a-united-states-office-of-marine-pollution-assessment-united-states-bureau-of-land-image232458103.htmlRMRE5AK3–. The Eastern Bering Sea Shelf : oceanography and resources / edited by Donald W. Hood and John A. Calder. Oceanography Bering Sea.. rT^R VII. 17^8 (H), 2l/3(H)-hopanes R= H, C2H5, CjHy VIII. extended 17a (H), 2l/3(H)-hopanes R-CH3-C5H11 405. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Hood, D. W. (Donald Wilbur), 1918-; Calder, John A; United States. Office of Marine Pollution Assessment; United States. Bureau of Land
 . Biochemistry of plants and animals, an introduction. Biochemistry. CH2 nCH -OH biliverdin (green) protein, forming biliverdin, a chain of pyrrole rings. This pigment is reduced to bilirubin (orange) which appears in the plasma in complexes with serum albumin and globulins. The liver traps the pigment and excretes it through the bile ducts into the duodenum of COOH COOH CHo CH2=CH CHg (CH2)2 (CH2)2 CHg CHg CH. bacteria bilirubin COOH COOH CHg C2H5 CHg (CH2)2 (CH2)2 CHg CHg C2H5 HO^- L-CH2-^ --0 ^- --'i'z. --0 —CH2-^ —a / ,,/ ^N"^ ^N^ ^N' ^N^ H H H H mesobiliru binogen OH. Please not Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemistry-of-plants-and-animals-an-introduction-biochemistry-ch2-nch-oh-biliverdin-green-protein-forming-biliverdin-a-chain-of-pyrrole-rings-this-pigment-is-reduced-to-bilirubin-orange-which-appears-in-the-plasma-in-complexes-with-serum-albumin-and-globulins-the-liver-traps-the-pigment-and-excretes-it-through-the-bile-ducts-into-the-duodenum-of-cooh-cooh-cho-ch2=ch-chg-ch22-ch22-chg-chg-ch-bacteria-bilirubin-cooh-cooh-chg-c2h5-chg-ch22-ch22-chg-chg-c2h5-ho-l-ch2-0-iz-0-ch2-a-nquot-n-n-n-h-h-h-h-mesobiliru-binogen-oh-please-not-image234677751.html
. Biochemistry of plants and animals, an introduction. Biochemistry. CH2 nCH -OH biliverdin (green) protein, forming biliverdin, a chain of pyrrole rings. This pigment is reduced to bilirubin (orange) which appears in the plasma in complexes with serum albumin and globulins. The liver traps the pigment and excretes it through the bile ducts into the duodenum of COOH COOH CHo CH2=CH CHg (CH2)2 (CH2)2 CHg CHg CH. bacteria bilirubin COOH COOH CHg C2H5 CHg (CH2)2 (CH2)2 CHg CHg C2H5 HO^- L-CH2-^ --0 ^- --'i'z. --0 —CH2-^ —a / ,,/ ^N"^ ^N^ ^N' ^N^ H H H H mesobiliru binogen OH. Please not Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/biochemistry-of-plants-and-animals-an-introduction-biochemistry-ch2-nch-oh-biliverdin-green-protein-forming-biliverdin-a-chain-of-pyrrole-rings-this-pigment-is-reduced-to-bilirubin-orange-which-appears-in-the-plasma-in-complexes-with-serum-albumin-and-globulins-the-liver-traps-the-pigment-and-excretes-it-through-the-bile-ducts-into-the-duodenum-of-cooh-cooh-cho-ch2=ch-chg-ch22-ch22-chg-chg-ch-bacteria-bilirubin-cooh-cooh-chg-c2h5-chg-ch22-ch22-chg-chg-c2h5-ho-l-ch2-0-iz-0-ch2-a-nquot-n-n-n-h-h-h-h-mesobiliru-binogen-oh-please-not-image234677751.htmlRMRHPDT7–. Biochemistry of plants and animals, an introduction. Biochemistry. CH2 nCH -OH biliverdin (green) protein, forming biliverdin, a chain of pyrrole rings. This pigment is reduced to bilirubin (orange) which appears in the plasma in complexes with serum albumin and globulins. The liver traps the pigment and excretes it through the bile ducts into the duodenum of COOH COOH CHo CH2=CH CHg (CH2)2 (CH2)2 CHg CHg CH. bacteria bilirubin COOH COOH CHg C2H5 CHg (CH2)2 (CH2)2 CHg CHg C2H5 HO^- L-CH2-^ --0 ^- --'i'z. --0 —CH2-^ —a / ,,/ ^N"^ ^N^ ^N' ^N^ H H H H mesobiliru binogen OH. Please not
 . dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/dan-bai-zhi-fen-zi-ji-chu-botany-2-6-m-so-ltch3coo-ltcrltbr-ltn03-ltc104-lti-ltcns-ch3n-ltnhzltrbknacsltliltmgtquotltca2ltba2-ch3inltc2h5ltnltc3h74nltchgn-sv-ltch3cooltcl-ltbrltc10cns-ch3nnhknaltliltca-ch3nltqh5nltc3hnltch8n-sds-image231730648.html
. dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/dan-bai-zhi-fen-zi-ji-chu-botany-2-6-m-so-ltch3coo-ltcrltbr-ltn03-ltc104-lti-ltcns-ch3n-ltnhzltrbknacsltliltmgtquotltca2ltba2-ch3inltc2h5ltnltc3h74nltchgn-sv-ltch3cooltcl-ltbrltc10cns-ch3nnhknaltliltca-ch3nltqh5nltc3hnltch8n-sds-image231730648.htmlRMRD06PG–. dan bai zhi fen zi ji chu. botany. 表â2åç§ç¦»å-对è6è´¨æ象稳å®æ§å½±åçç¸å¯¹å¤§å° èºæ -天ç¶æ象+ çæ æ è§å·æ² åæ§ç¶æ- çm è¶å-æè¶ SO,^-<CH3COO-<Cr<Br-<N03-<C104-<I-<CNS (CH3ãN + <NHZ<Rb+ï¼K+,Na+,Cs+<Liâ<Mg:t"<Ca2+<Ba2+ (CH3),iN+<(C2H5)<N+<(C3H7)4N+<(C,HgãN+. S(V-<CH3COOâ<Cl-<Brâ<C10^ãCNS- (CH3)«N+,NH,+,K+,Na+<Li+<Ca^+ (CH3),N+<(QH5),N+<(C3H,),N+<(C,H8),N + ååºï¼å¸¸å¸¸å¼èµ·èç½è´¨è§£ç¦»æäºåºæå个è½é¾çåæ§ãä¸è²ãçé ¸èä¸åï¼è¿ç±»ç©è´¨ä¸éè¦é«æµ 度ï¼å¨å¾ä½çæµåº¦ä¸ï¼å°±è½åèç½è´¨é«åº¦å°ç»åã SDSä¸èç½è´¨çç¸äºä½ç¨æå¾å¤ç¨é
 . Die wissenschaftliche Ausbildung des Apothekerlehrlings und seine Vorbereitung zum Gehülfenexamen : mit Rücksicht auf die neuesten Anforderungen. Pharmacy; Chemistry; Physics; Botany; Education, Pharmacy. - 263. Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Sc Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/die-wissenschaftliche-ausbildung-des-apothekerlehrlings-und-seine-vorbereitung-zum-gehlfenexamen-mit-rcksicht-auf-die-neuesten-anforderungen-pharmacy-chemistry-physics-botany-education-pharmacy-263-fig-71-theorie-der-therbildung-der-ther-stellt-das-oxyd-des-im-quotweingeist-enthaltenen-thvls-c2h5-dar-c4h1no-=-c2h520-ther-thyloxyd-wie-geht-nun-der-weingeist-thylhydroxyd-in-ther-thyloxyd-ber-frher-glaubte-man-dass-die-konz-schwefel-sure-vermge-ihrer-wasseranziehenden-kraft-dem-weingeist-hydratwasser-entzge-da-aber-die-wasseranziehende-kraft-der-sc-image231399687.html
. Die wissenschaftliche Ausbildung des Apothekerlehrlings und seine Vorbereitung zum Gehülfenexamen : mit Rücksicht auf die neuesten Anforderungen. Pharmacy; Chemistry; Physics; Botany; Education, Pharmacy. - 263. Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Sc Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/die-wissenschaftliche-ausbildung-des-apothekerlehrlings-und-seine-vorbereitung-zum-gehlfenexamen-mit-rcksicht-auf-die-neuesten-anforderungen-pharmacy-chemistry-physics-botany-education-pharmacy-263-fig-71-theorie-der-therbildung-der-ther-stellt-das-oxyd-des-im-quotweingeist-enthaltenen-thvls-c2h5-dar-c4h1no-=-c2h520-ther-thyloxyd-wie-geht-nun-der-weingeist-thylhydroxyd-in-ther-thyloxyd-ber-frher-glaubte-man-dass-die-konz-schwefel-sure-vermge-ihrer-wasseranziehenden-kraft-dem-weingeist-hydratwasser-entzge-da-aber-die-wasseranziehende-kraft-der-sc-image231399687.htmlRMRCD4JF–. Die wissenschaftliche Ausbildung des Apothekerlehrlings und seine Vorbereitung zum Gehülfenexamen : mit Rücksicht auf die neuesten Anforderungen. Pharmacy; Chemistry; Physics; Botany; Education, Pharmacy. - 263. Fig. 71. Theorie der Ätherbildung. Der Äther stellt das Oxyd des im "Weingeist enthaltenen Äthvls (C2H5) dar: C4H1nO = (C2H5)20 Äther Äthyloxyd. Wie geht nun der Weingeist (Äthylhydroxyd) in Äther (Äthyloxyd) über? Früher glaubte man, dass die konz. Schwefel- säure vermöge ihrer wasseranziehenden Kraft dem Weingeist Hydratwasser entzöge; da aber die wasseranziehende Kraft der Sc
Search Results for C2h5 Stock Photos and Images (31)
Page 1 of 1