Quick filters:
Page 1 of 1
Sodium thiosulphate Stock Photos and Images
 Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563370042.html
Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563370042.htmlRF2RMFM6J–Sodium thiosulfate or sodium thiosulphate on blue background.
 Test tube spilling Sodium thiosulphate Na2S2O3 colourless crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-test-tube-spilling-sodium-thiosulphate-na2s2o3-colourless-crystals-54655762.html
Test tube spilling Sodium thiosulphate Na2S2O3 colourless crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-test-tube-spilling-sodium-thiosulphate-na2s2o3-colourless-crystals-54655762.htmlRMD4WNYE–Test tube spilling Sodium thiosulphate Na2S2O3 colourless crystals
 A bottle of one molar 1M sodium thiosulphate liquid to be used for bromine spillage, as used in a UK secondary school, London, UK. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-bottle-of-one-molar-1m-sodium-thiosulphate-liquid-to-be-used-for-bromine-spillage-as-used-in-a-uk-secondary-school-london-uk-image178884513.html
A bottle of one molar 1M sodium thiosulphate liquid to be used for bromine spillage, as used in a UK secondary school, London, UK. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-bottle-of-one-molar-1m-sodium-thiosulphate-liquid-to-be-used-for-bromine-spillage-as-used-in-a-uk-secondary-school-london-uk-image178884513.htmlRMMB0W0H–A bottle of one molar 1M sodium thiosulphate liquid to be used for bromine spillage, as used in a UK secondary school, London, UK.
 Pile of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-pile-of-sodium-thiosulphate-35120098.html
Pile of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-pile-of-sodium-thiosulphate-35120098.htmlRMC13T16–Pile of Sodium Thiosulphate
 sodium thiosulphate reacts with HCl & as sulphur is precipitated the cross gradually disappears Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-reacts-with-hcl-as-sulphur-is-precipitated-the-19455226.html
sodium thiosulphate reacts with HCl & as sulphur is precipitated the cross gradually disappears Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-reacts-with-hcl-as-sulphur-is-precipitated-the-19455226.htmlRMB3J79E–sodium thiosulphate reacts with HCl & as sulphur is precipitated the cross gradually disappears
 Sodium thiosulphate intravenous solution, conceptual image. Neutralizes cyanide toxicity and treats calciphylaxis in dialysis patients. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulphate-intravenous-solution-conceptual-image-neutralizes-cyanide-toxicity-and-treats-calciphylaxis-in-dialysis-patients-image601415656.html
Sodium thiosulphate intravenous solution, conceptual image. Neutralizes cyanide toxicity and treats calciphylaxis in dialysis patients. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulphate-intravenous-solution-conceptual-image-neutralizes-cyanide-toxicity-and-treats-calciphylaxis-in-dialysis-patients-image601415656.htmlRF2WXCRPG–Sodium thiosulphate intravenous solution, conceptual image. Neutralizes cyanide toxicity and treats calciphylaxis in dialysis patients.
 Microscale rate of reaction experiment between sodium thiosulphate and hydrochloric acid Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/microscale-rate-of-reaction-experiment-between-sodium-thiosulphate-and-hydrochloric-acid-image232865406.html
Microscale rate of reaction experiment between sodium thiosulphate and hydrochloric acid Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/microscale-rate-of-reaction-experiment-between-sodium-thiosulphate-and-hydrochloric-acid-image232865406.htmlRFRERX5J–Microscale rate of reaction experiment between sodium thiosulphate and hydrochloric acid
 Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-120356124.html
Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-120356124.htmlRFGYPKD0–Sodium thiosulphate crystals in polarized light
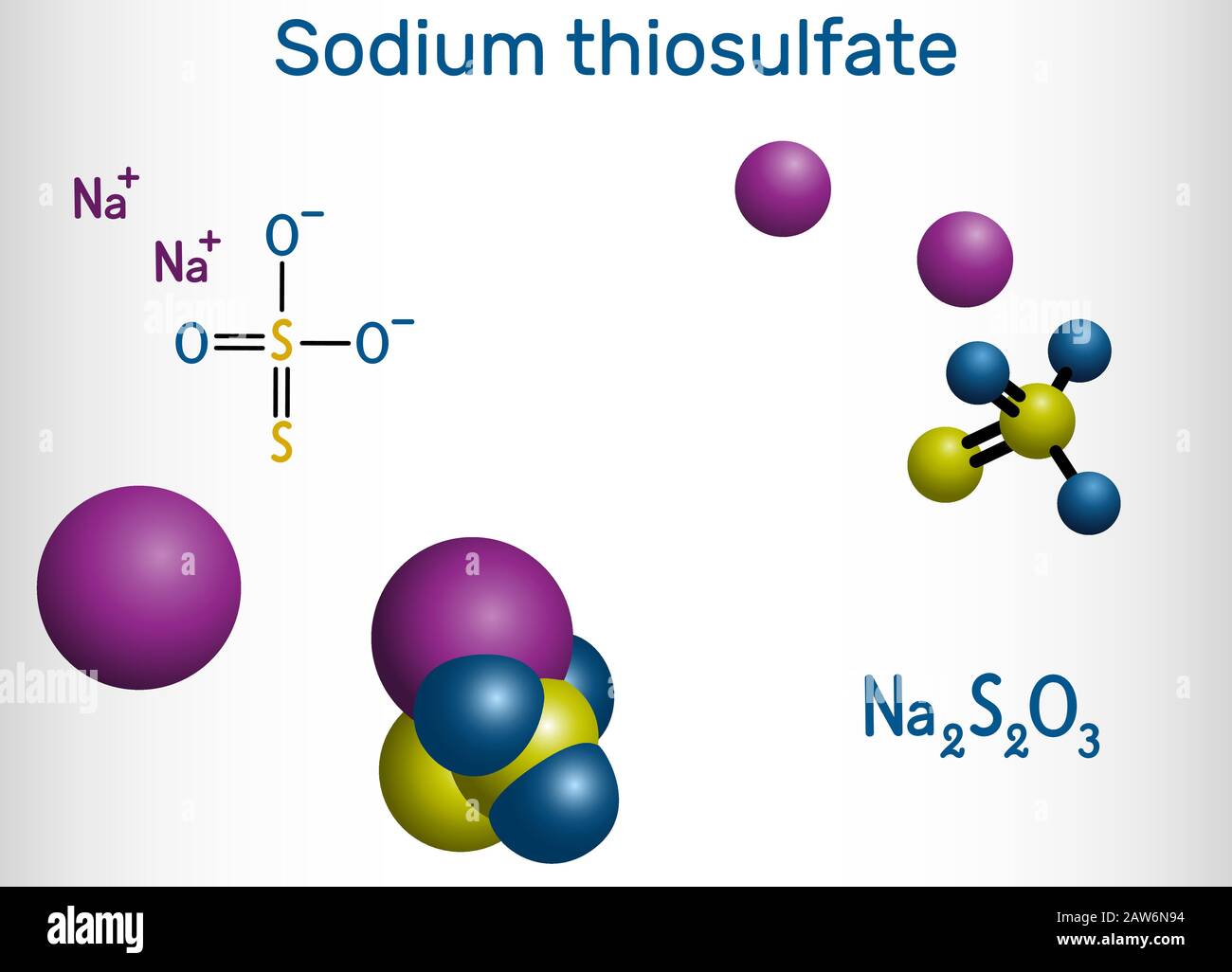 Sodium thiosulfate, sodium thiosulphate, Na2S2O3 molecule. It is used to treat cyanide poisoning, pityriasis versicolor, and to decrease side effects Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-sodium-thiosulphate-na2s2o3-molecule-it-is-used-to-treat-cyanide-poisoning-pityriasis-versicolor-and-to-decrease-side-effects-image342511824.html
Sodium thiosulfate, sodium thiosulphate, Na2S2O3 molecule. It is used to treat cyanide poisoning, pityriasis versicolor, and to decrease side effects Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-sodium-thiosulphate-na2s2o3-molecule-it-is-used-to-treat-cyanide-poisoning-pityriasis-versicolor-and-to-decrease-side-effects-image342511824.htmlRF2AW6N94–Sodium thiosulfate, sodium thiosulphate, Na2S2O3 molecule. It is used to treat cyanide poisoning, pityriasis versicolor, and to decrease side effects
 Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-116592816.html
Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-116592816.htmlRFGNK794–Sodium thiosulphate crystals in polarized light
 Container of the food additive E539 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/container-of-the-food-additive-e539-image486038094.html
Container of the food additive E539 Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/container-of-the-food-additive-e539-image486038094.htmlRF2K6MXFA–Container of the food additive E539
 Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-115246837.html
Sodium thiosulphate crystals in polarized light Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-sodium-thiosulphate-crystals-in-polarized-light-115246837.htmlRFGKDXED–Sodium thiosulphate crystals in polarized light
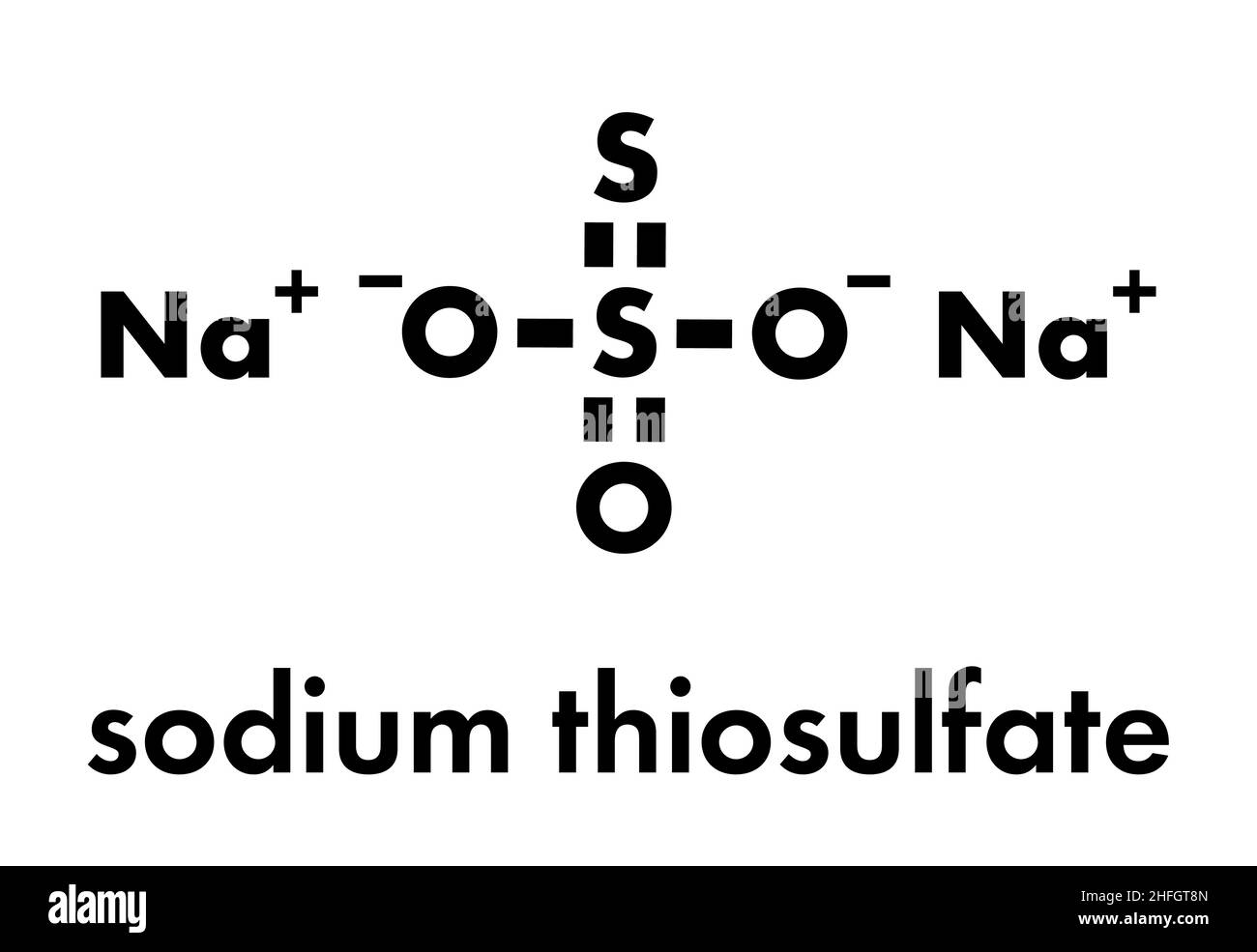 Sodium thiosulfate (hyposulfite) salt, chemical structure. Skeletal formula. Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-skeletal-formula-image457059701.html
Sodium thiosulfate (hyposulfite) salt, chemical structure. Skeletal formula. Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-skeletal-formula-image457059701.htmlRF2HFGT8N–Sodium thiosulfate (hyposulfite) salt, chemical structure. Skeletal formula.
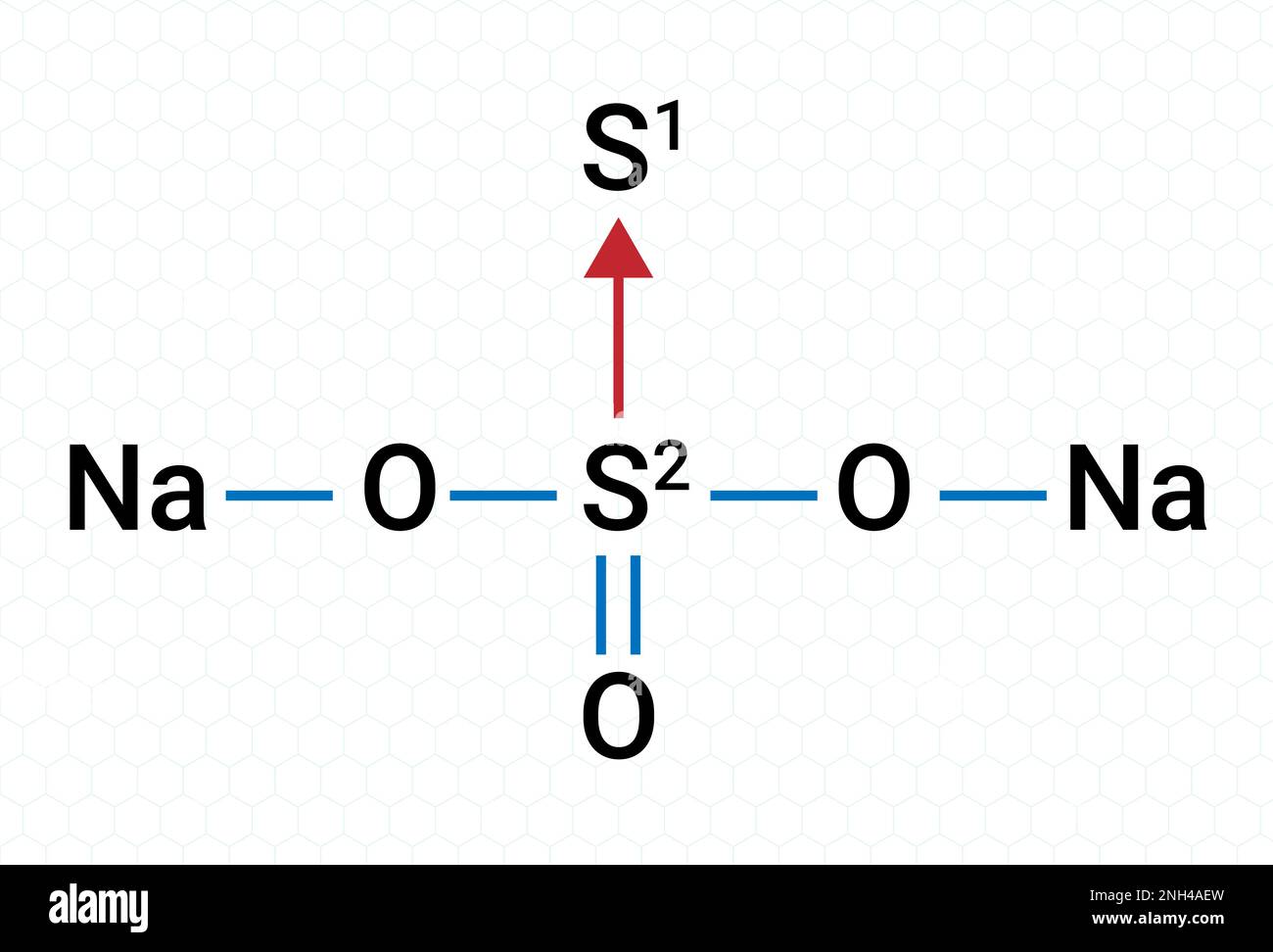 Chemical Structure of Na2S2O3 (Sodium thiosulfate) Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemical-structure-of-na2s2o3-sodium-thiosulfate-image526856257.html
Chemical Structure of Na2S2O3 (Sodium thiosulfate) Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/chemical-structure-of-na2s2o3-sodium-thiosulfate-image526856257.htmlRF2NH4AEW–Chemical Structure of Na2S2O3 (Sodium thiosulfate)
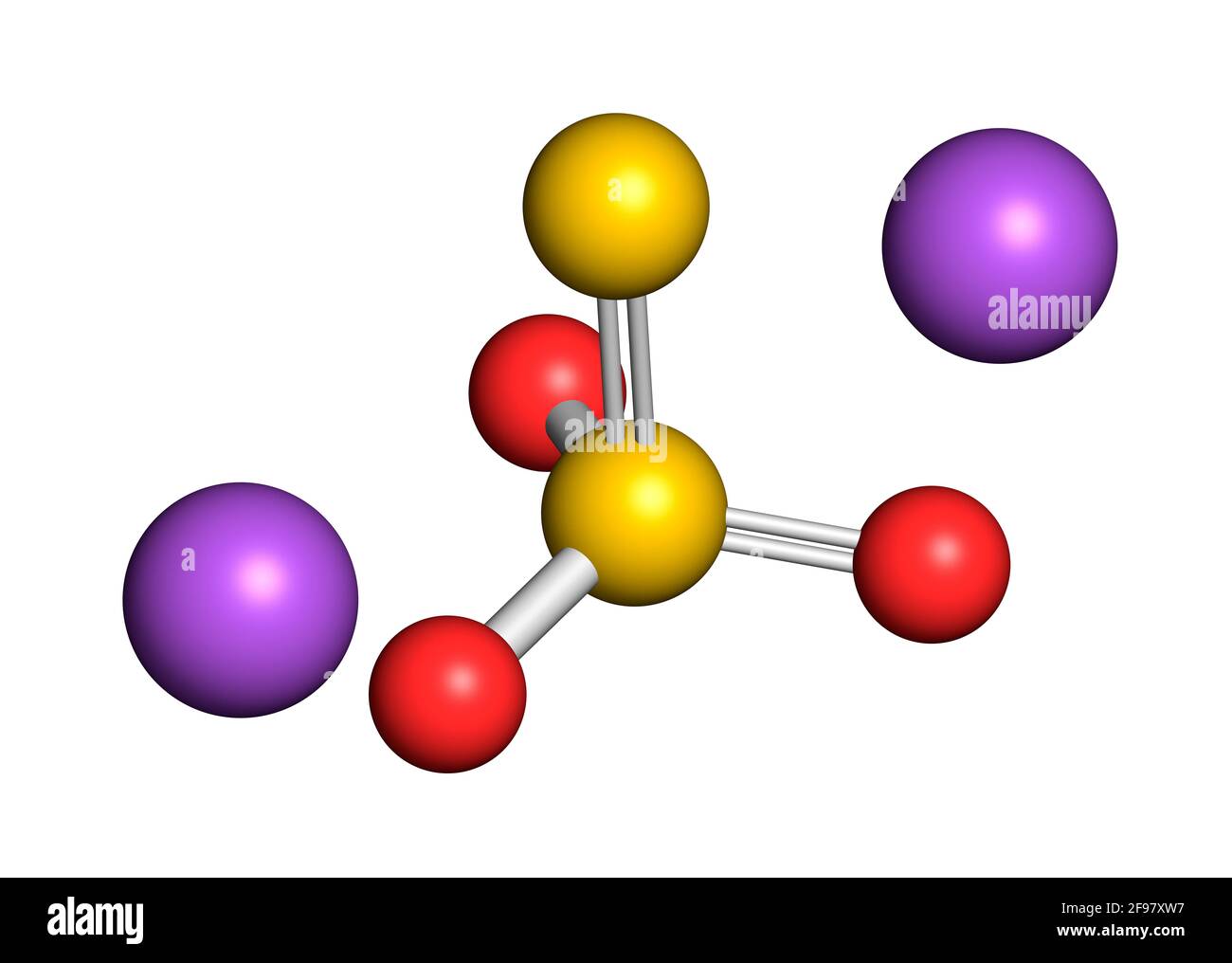 Sodium thiosulfate chemical structure, illustration Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-illustration-image418755491.html
Sodium thiosulfate chemical structure, illustration Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-illustration-image418755491.htmlRF2F97XW7–Sodium thiosulfate chemical structure, illustration
 Medical Syringe. Injection. Treatment Drugs Syringe. Medicine. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/medical-syringe-injection-treatment-drugs-syringe-medicine-image225635292.html
Medical Syringe. Injection. Treatment Drugs Syringe. Medicine. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/medical-syringe-injection-treatment-drugs-syringe-medicine-image225635292.htmlRFR32G38–Medical Syringe. Injection. Treatment Drugs Syringe. Medicine.
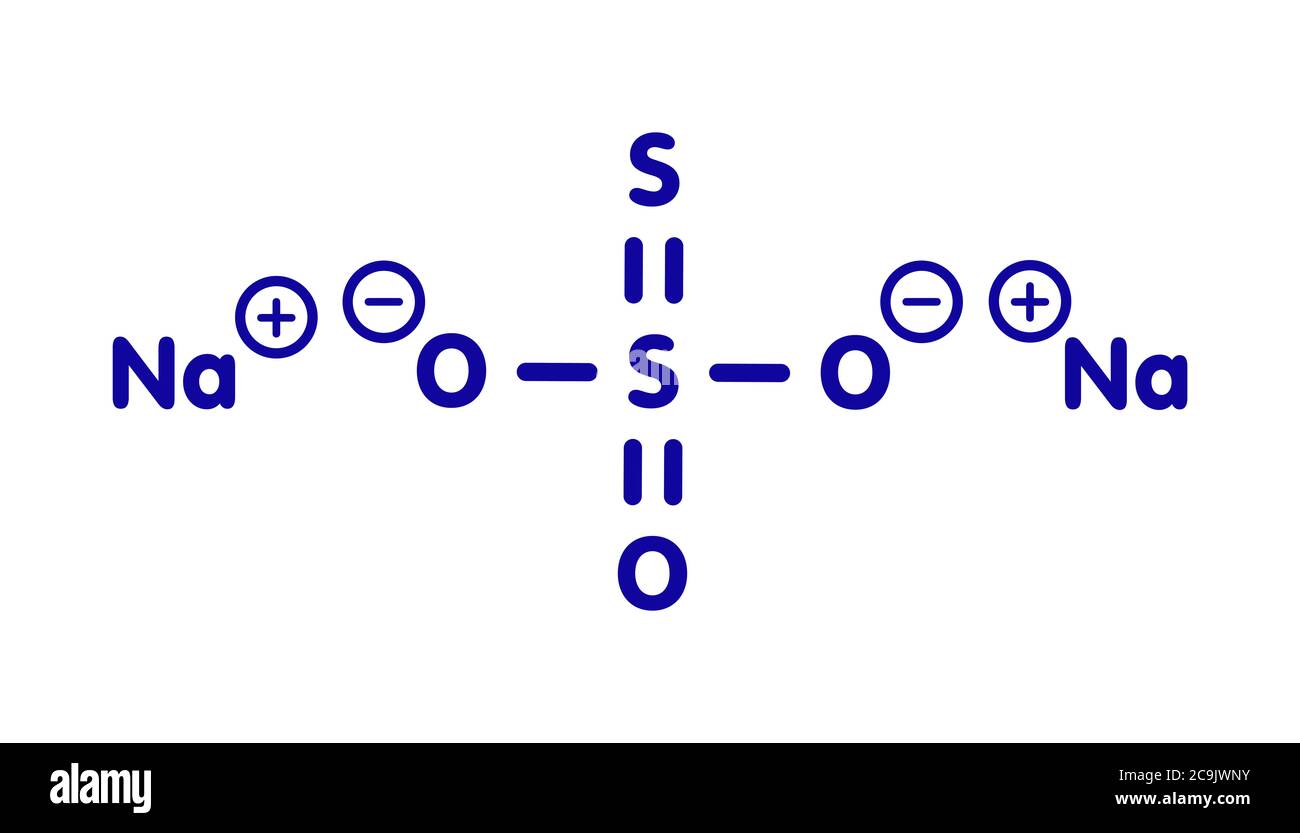 Sodium thiosulfate (hyposulfite) salt, chemical structure. Blue skeletal formula on white background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-blue-skeletal-formula-on-white-background-image367364983.html
Sodium thiosulfate (hyposulfite) salt, chemical structure. Blue skeletal formula on white background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-blue-skeletal-formula-on-white-background-image367364983.htmlRF2C9JWNY–Sodium thiosulfate (hyposulfite) salt, chemical structure. Blue skeletal formula on white background.
 Sodium thiosulfate, chemical structure. Skeletal formula. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-skeletal-formula-image235249781.html
Sodium thiosulfate, chemical structure. Skeletal formula. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-skeletal-formula-image235249781.htmlRFRJMFDW–Sodium thiosulfate, chemical structure. Skeletal formula.
 Close-Up of Sodium Thiosulphate Crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-sodium-thiosulphate-crystals-19815784.html
Close-Up of Sodium Thiosulphate Crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-close-up-of-sodium-thiosulphate-crystals-19815784.htmlRFB46K6G–Close-Up of Sodium Thiosulphate Crystals
 Sodium thiosulfate or sodium thiosulphate on beige background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-beige-background-image563370525.html
Sodium thiosulfate or sodium thiosulphate on beige background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-beige-background-image563370525.htmlRF2RMFMRW–Sodium thiosulfate or sodium thiosulphate on beige background.
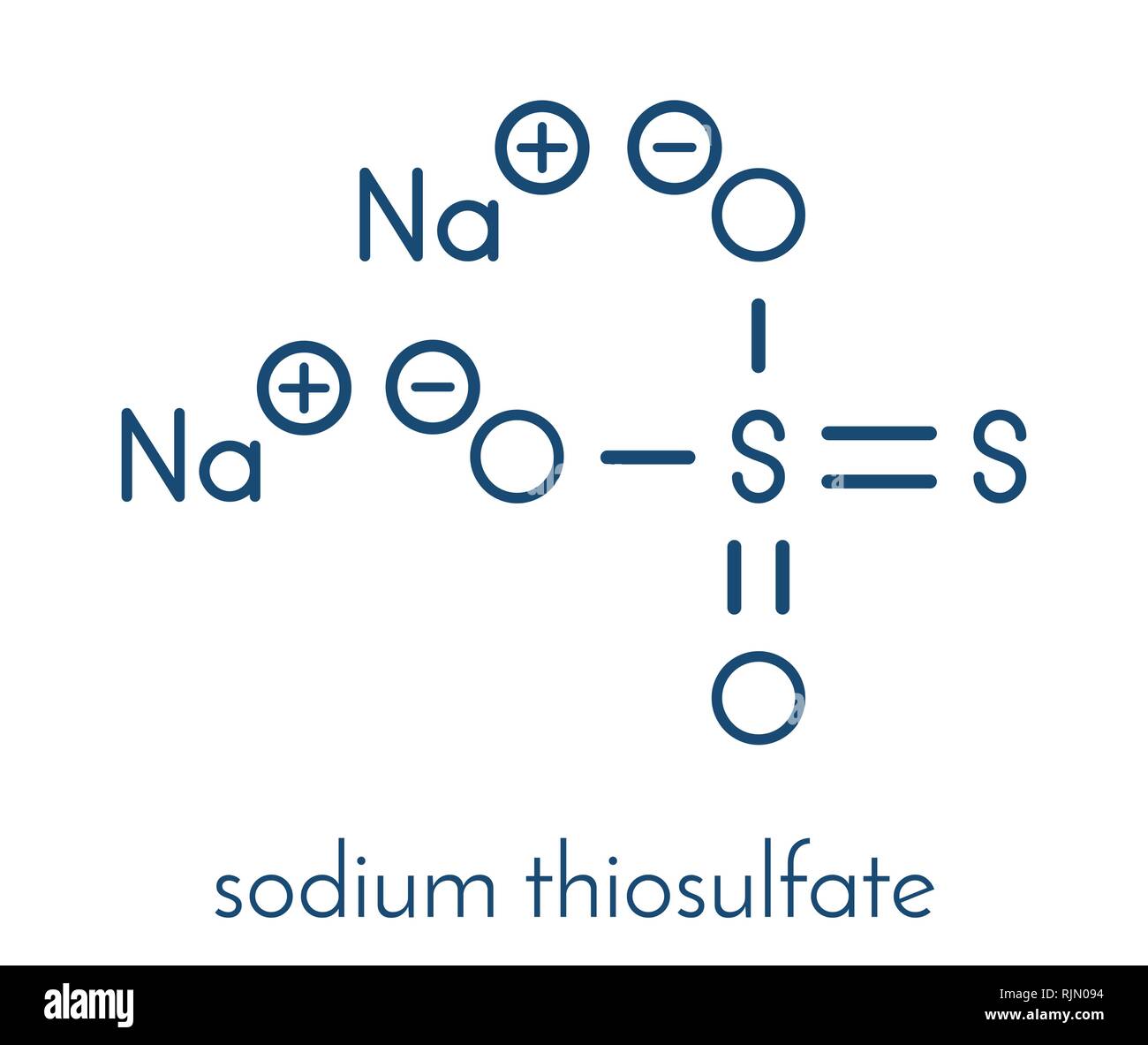 Sodium thiosulfate, chemical structure. Skeletal formula. Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-skeletal-formula-image235259840.html
Sodium thiosulfate, chemical structure. Skeletal formula. Stock Vectorhttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-skeletal-formula-image235259840.htmlRFRJN094–Sodium thiosulfate, chemical structure. Skeletal formula.
 redoxtitratie kaliumpermanganaat met natriumthiosulfaat in erlenmeyer met vlo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/redoxtitratie-kaliumpermanganaat-met-natriumthiosulfaat-in-erlenmeyer-met-vlo-image466465521.html
redoxtitratie kaliumpermanganaat met natriumthiosulfaat in erlenmeyer met vlo Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/redoxtitratie-kaliumpermanganaat-met-natriumthiosulfaat-in-erlenmeyer-met-vlo-image466465521.htmlRM2J2W9EW–redoxtitratie kaliumpermanganaat met natriumthiosulfaat in erlenmeyer met vlo
 Opened plastic container of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-opened-plastic-container-of-sodium-thiosulphate-35119864.html
Opened plastic container of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-opened-plastic-container-of-sodium-thiosulphate-35119864.htmlRMC13RMT–Opened plastic container of Sodium Thiosulphate
 Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which has almost obscured x Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-rate-of-reaction-experiment-in-which-sodium-thiosulphate-and-hydrochloric-16775967.html
Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which has almost obscured x Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-rate-of-reaction-experiment-in-which-sodium-thiosulphate-and-hydrochloric-16775967.htmlRMAX9R4G–Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which has almost obscured x
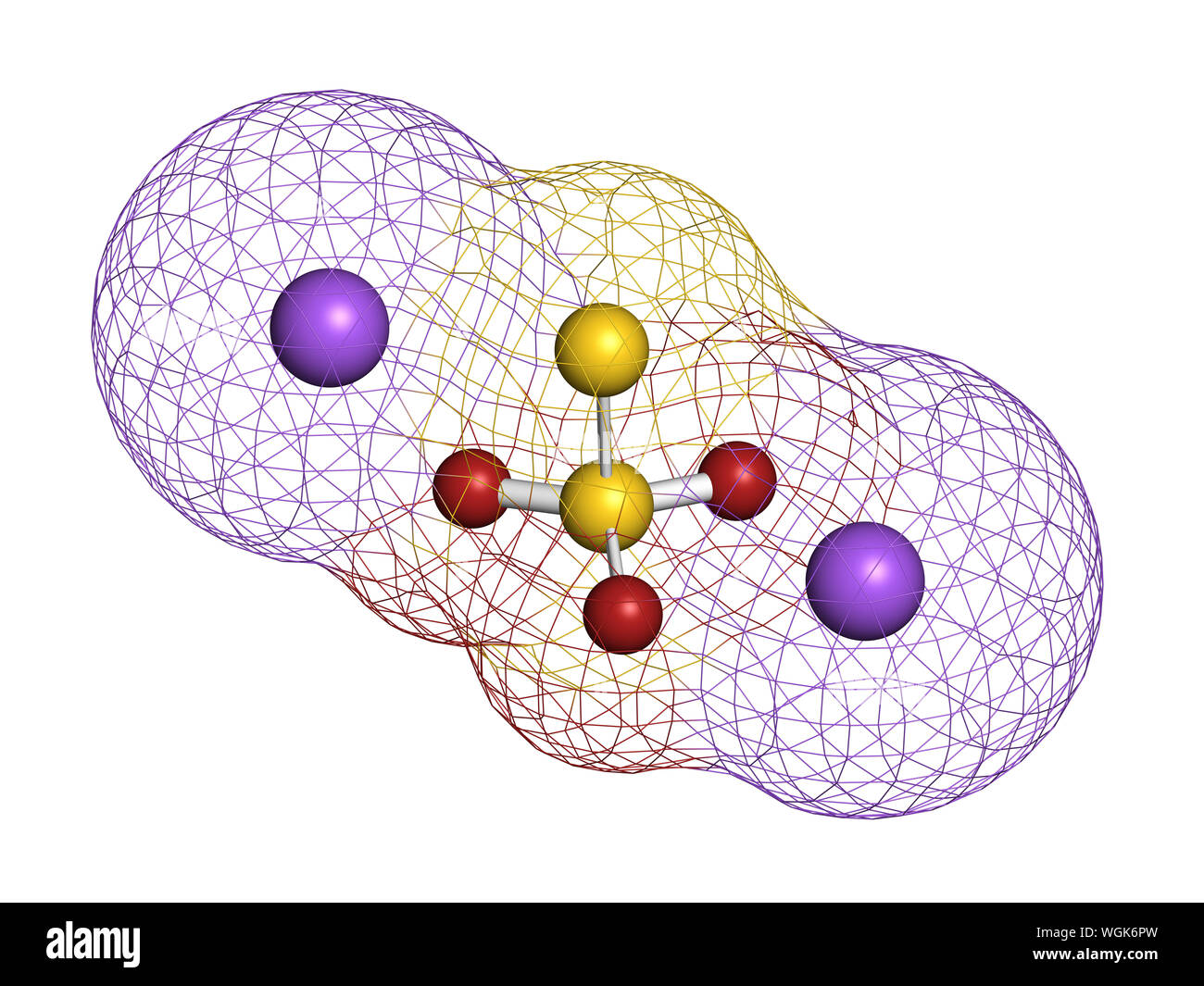 Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412449.html
Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412449.htmlRFWGK6PW–Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red),
 Glass dish with suspension of sulphur in sodium sulphite heated on a tripod. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/glass-dish-with-suspension-of-sulphur-in-sodium-sulphite-heated-on-a-tripod-image216107444.html
Glass dish with suspension of sulphur in sodium sulphite heated on a tripod. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/glass-dish-with-suspension-of-sulphur-in-sodium-sulphite-heated-on-a-tripod-image216107444.htmlRMPFGF70–Glass dish with suspension of sulphur in sodium sulphite heated on a tripod.
![. Journal. i dh, ii duced ; tr. a I men I with potassium bii Inornate(2 mols.) and sodium thiosulphate (1 mol.); ti atmenfchromic acid and sodium nitrite; treatmnth potassium chromate (1 mol.) and a basicchromic salt (1 mol.). Instead of using excess of hydro-i ic acid in the dyeing process, nitric or sulphuric acidsmay be used, and a part of thi mineral acid may beadvantageously replaced by an organic acid (particularlyformic or oxalic acid). The dyeing] may be carried out ,by immersing the mordanted fibre in the acid solutionof aniline for a short time, ami then removing it, andallowing the Stock Photo . Journal. i dh, ii duced ; tr. a I men I with potassium bii Inornate(2 mols.) and sodium thiosulphate (1 mol.); ti atmenfchromic acid and sodium nitrite; treatmnth potassium chromate (1 mol.) and a basicchromic salt (1 mol.). Instead of using excess of hydro-i ic acid in the dyeing process, nitric or sulphuric acidsmay be used, and a part of thi mineral acid may beadvantageously replaced by an organic acid (particularlyformic or oxalic acid). The dyeing] may be carried out ,by immersing the mordanted fibre in the acid solutionof aniline for a short time, ami then removing it, andallowing the Stock Photo](https://c8.alamy.com/comp/2AG8T3H/journal-i-dh-ii-duced-tr-a-i-men-i-with-potassium-bii-inornate2-mols-and-sodium-thiosulphate-1-mol-ti-atmenfchromic-acid-and-sodium-nitrite-treatmnth-potassium-chromate-1-mol-and-a-basicchromic-salt-1-mol-instead-of-using-excess-of-hydro-i-ic-acid-in-the-dyeing-process-nitric-or-sulphuric-acidsmay-be-used-and-a-part-of-thi-mineral-acid-may-beadvantageously-replaced-by-an-organic-acid-particularlyformic-or-oxalic-acid-the-dyeing-may-be-carried-out-by-immersing-the-mordanted-fibre-in-the-acid-solutionof-aniline-for-a-short-time-ami-then-removing-it-andallowing-the-2AG8T3H.jpg) . Journal. i dh, ii duced ; tr. a I men I with potassium bii Inornate(2 mols.) and sodium thiosulphate (1 mol.); ti atmenfchromic acid and sodium nitrite; treatmnth potassium chromate (1 mol.) and a basicchromic salt (1 mol.). Instead of using excess of hydro-i ic acid in the dyeing process, nitric or sulphuric acidsmay be used, and a part of thi mineral acid may beadvantageously replaced by an organic acid (particularlyformic or oxalic acid). The dyeing] may be carried out ,by immersing the mordanted fibre in the acid solutionof aniline for a short time, ami then removing it, andallowing the Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/journal-i-dh-ii-duced-tr-a-i-men-i-with-potassium-bii-inornate2-mols-and-sodium-thiosulphate-1-mol-ti-atmenfchromic-acid-and-sodium-nitrite-treatmnth-potassium-chromate-1-mol-and-a-basicchromic-salt-1-mol-instead-of-using-excess-of-hydro-i-ic-acid-in-the-dyeing-process-nitric-or-sulphuric-acidsmay-be-used-and-a-part-of-thi-mineral-acid-may-beadvantageously-replaced-by-an-organic-acid-particularlyformic-or-oxalic-acid-the-dyeing-may-be-carried-out-by-immersing-the-mordanted-fibre-in-the-acid-solutionof-aniline-for-a-short-time-ami-then-removing-it-andallowing-the-image337026021.html
. Journal. i dh, ii duced ; tr. a I men I with potassium bii Inornate(2 mols.) and sodium thiosulphate (1 mol.); ti atmenfchromic acid and sodium nitrite; treatmnth potassium chromate (1 mol.) and a basicchromic salt (1 mol.). Instead of using excess of hydro-i ic acid in the dyeing process, nitric or sulphuric acidsmay be used, and a part of thi mineral acid may beadvantageously replaced by an organic acid (particularlyformic or oxalic acid). The dyeing] may be carried out ,by immersing the mordanted fibre in the acid solutionof aniline for a short time, ami then removing it, andallowing the Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/journal-i-dh-ii-duced-tr-a-i-men-i-with-potassium-bii-inornate2-mols-and-sodium-thiosulphate-1-mol-ti-atmenfchromic-acid-and-sodium-nitrite-treatmnth-potassium-chromate-1-mol-and-a-basicchromic-salt-1-mol-instead-of-using-excess-of-hydro-i-ic-acid-in-the-dyeing-process-nitric-or-sulphuric-acidsmay-be-used-and-a-part-of-thi-mineral-acid-may-beadvantageously-replaced-by-an-organic-acid-particularlyformic-or-oxalic-acid-the-dyeing-may-be-carried-out-by-immersing-the-mordanted-fibre-in-the-acid-solutionof-aniline-for-a-short-time-ami-then-removing-it-andallowing-the-image337026021.htmlRM2AG8T3H–. Journal. i dh, ii duced ; tr. a I men I with potassium bii Inornate(2 mols.) and sodium thiosulphate (1 mol.); ti atmenfchromic acid and sodium nitrite; treatmnth potassium chromate (1 mol.) and a basicchromic salt (1 mol.). Instead of using excess of hydro-i ic acid in the dyeing process, nitric or sulphuric acidsmay be used, and a part of thi mineral acid may beadvantageously replaced by an organic acid (particularlyformic or oxalic acid). The dyeing] may be carried out ,by immersing the mordanted fibre in the acid solutionof aniline for a short time, ami then removing it, andallowing the
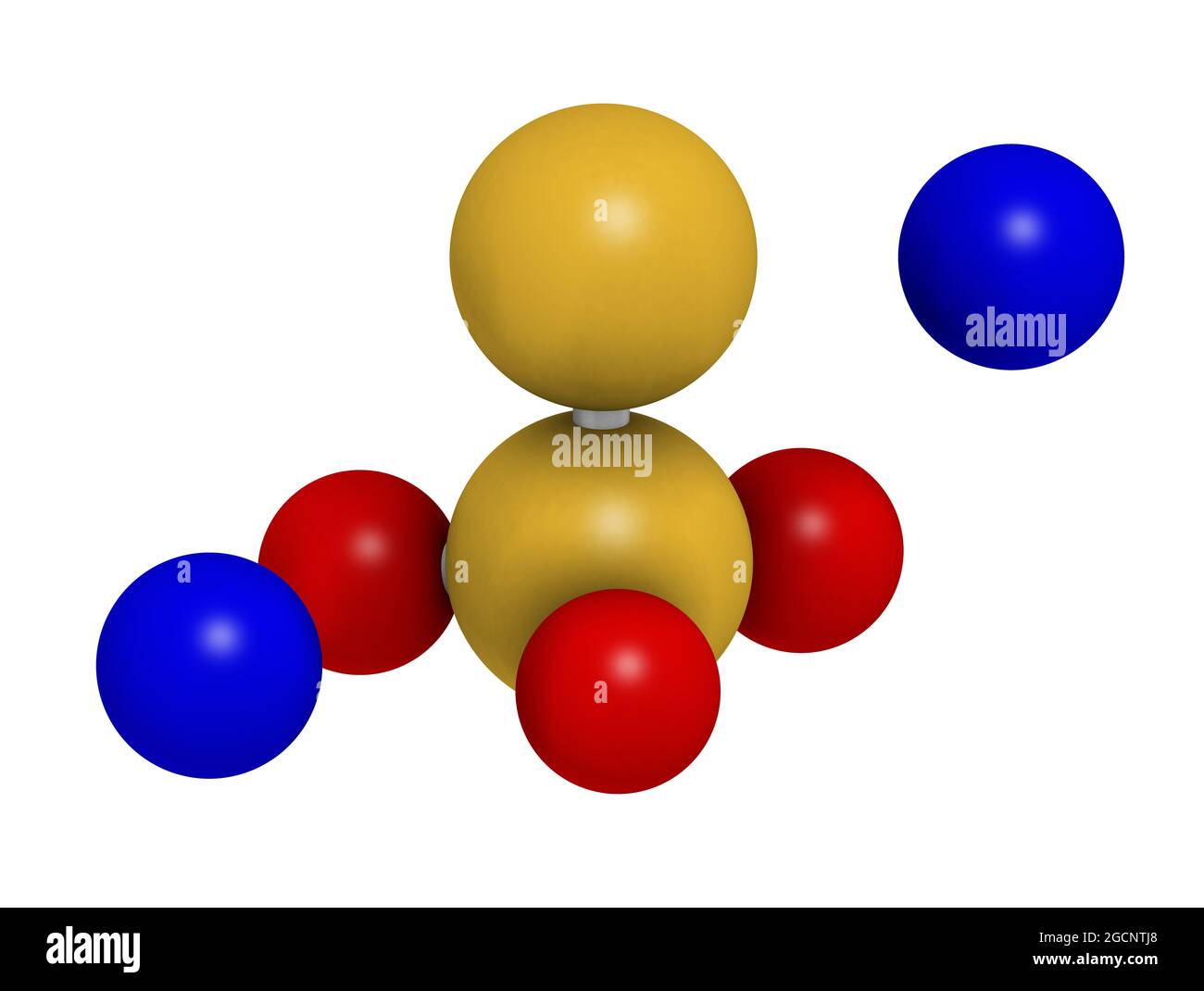 Sodium thiosulfate, chemical structure. 3D rendering. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-image438115392.html
Sodium thiosulfate, chemical structure. 3D rendering. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-image438115392.htmlRF2GCNTJ8–Sodium thiosulfate, chemical structure. 3D rendering.
 Medical Syringe. Injection. Treatment Drugs Syringe. Hormone. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/medical-syringe-injection-treatment-drugs-syringe-hormone-image225635293.html
Medical Syringe. Injection. Treatment Drugs Syringe. Hormone. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/medical-syringe-injection-treatment-drugs-syringe-hormone-image225635293.htmlRFR32G39–Medical Syringe. Injection. Treatment Drugs Syringe. Hormone.
 Sodium thiosulfate (hyposulfite) salt, chemical structure. White skeletal formula on dark teal gradient background with hexagonal pattern. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-white-skeletal-formula-on-dark-teal-gradient-background-with-hexagonal-pattern-image367364958.html
Sodium thiosulfate (hyposulfite) salt, chemical structure. White skeletal formula on dark teal gradient background with hexagonal pattern. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-white-skeletal-formula-on-dark-teal-gradient-background-with-hexagonal-pattern-image367364958.htmlRF2C9JWN2–Sodium thiosulfate (hyposulfite) salt, chemical structure. White skeletal formula on dark teal gradient background with hexagonal pattern.
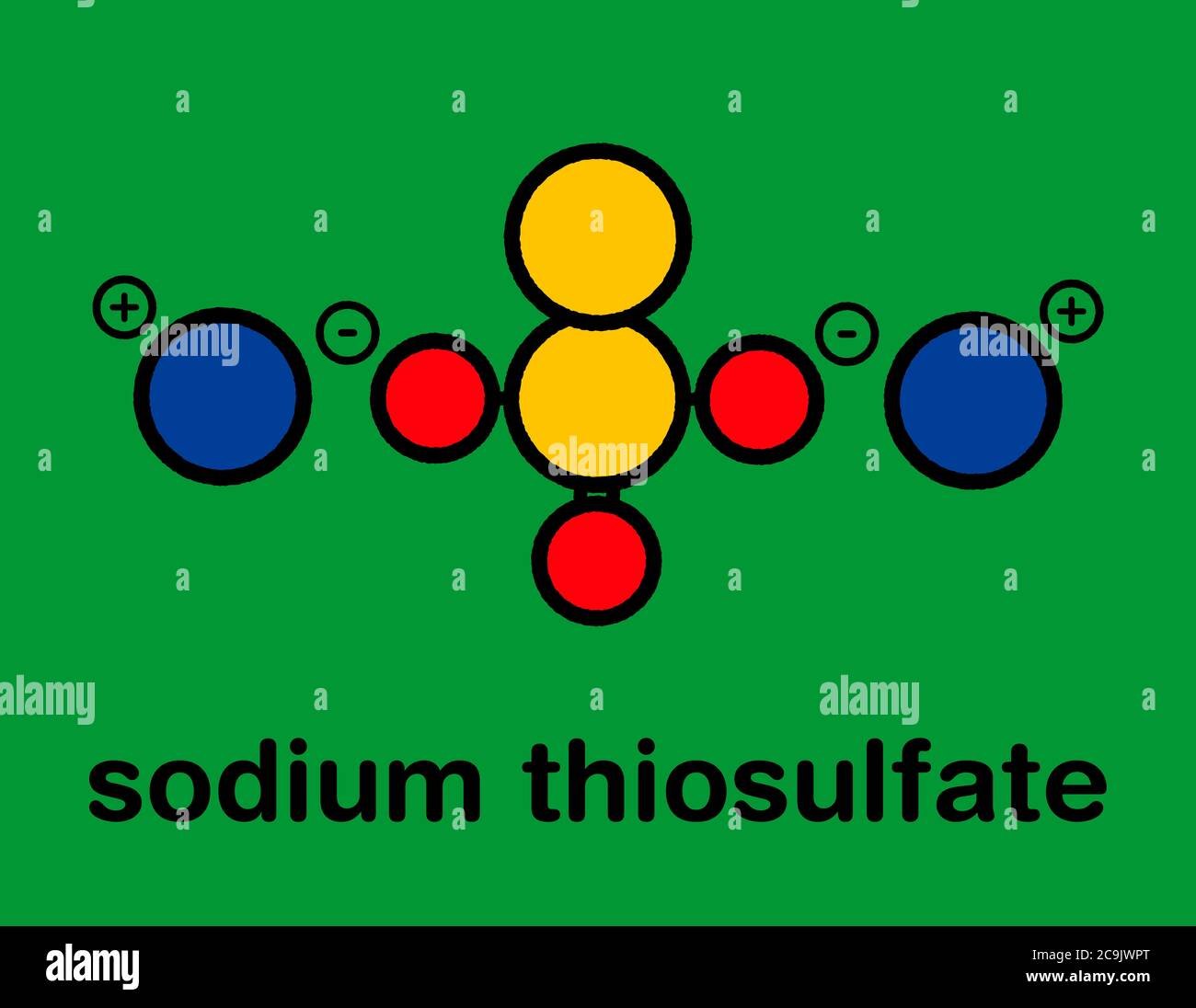 Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles with Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-stylized-skeletal-formula-chemical-structure-atoms-are-shown-as-color-coded-circles-with-image367365008.html
Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles with Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-stylized-skeletal-formula-chemical-structure-atoms-are-shown-as-color-coded-circles-with-image367365008.htmlRF2C9JWPT–Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles with
 Sodium thiosulfate or sodium thiosulphate on beige background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-beige-background-image563370341.html
Sodium thiosulfate or sodium thiosulphate on beige background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-beige-background-image563370341.htmlRF2RMFMH9–Sodium thiosulfate or sodium thiosulphate on beige background.
 Plastic container containing Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-plastic-container-containing-sodium-thiosulphate-35119743.html
Plastic container containing Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-plastic-container-containing-sodium-thiosulphate-35119743.htmlRMC13RGF–Plastic container containing Sodium Thiosulphate
 Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which eventually obscures x Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-rate-of-reaction-experiment-in-which-sodium-thiosulphate-and-hydrochloric-16774456.html
Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which eventually obscures x Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-rate-of-reaction-experiment-in-which-sodium-thiosulphate-and-hydrochloric-16774456.htmlRMAX9JJH–Rate of reaction experiment in which sodium thiosulphate and hydrochloric acid react to form sulphur which eventually obscures x
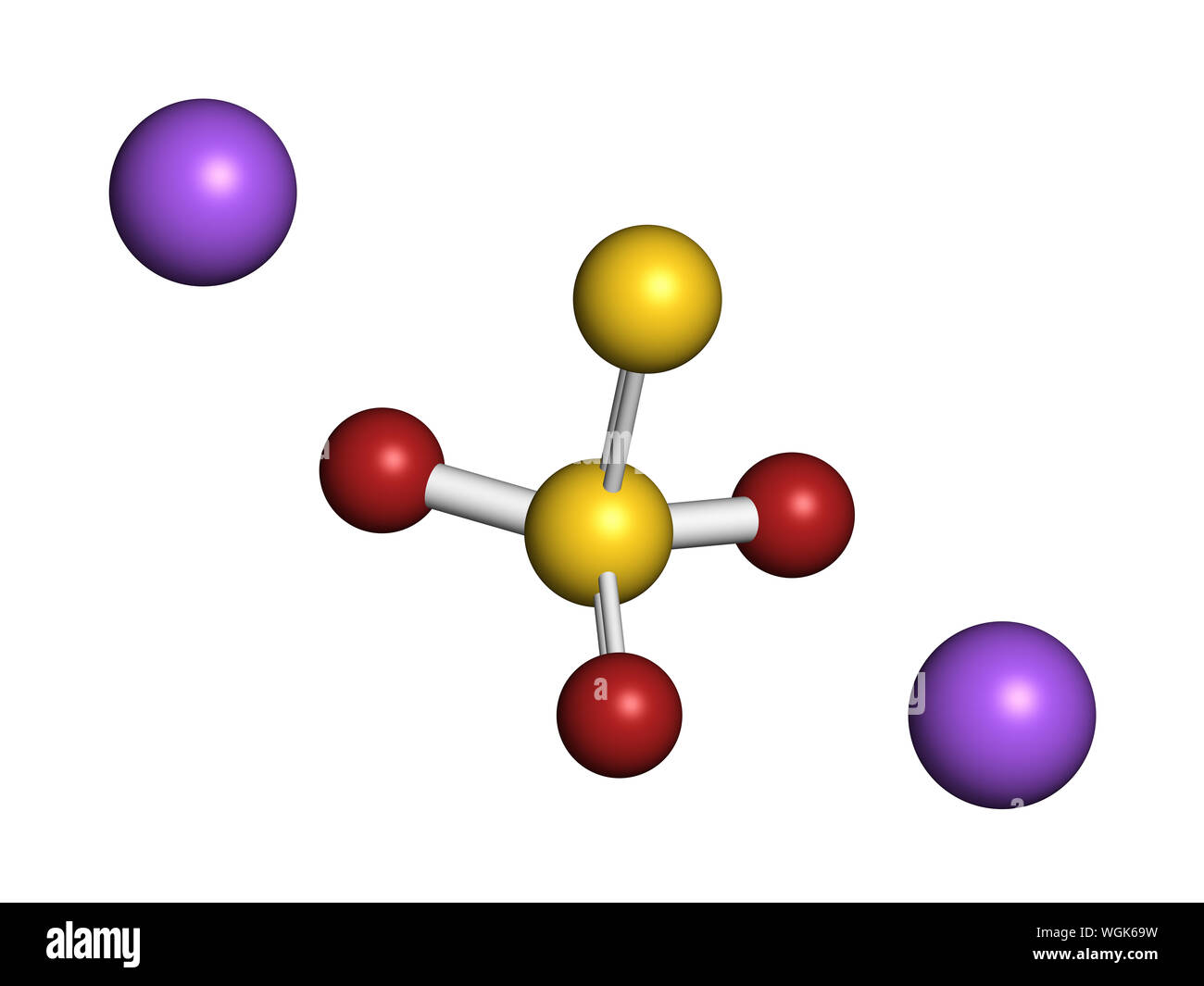 Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412085.html
Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412085.htmlRFWGK69W–Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red),
 . Hydroxylamine : its quantitative determination and some double salts . ne solu-tion and titrate back with sodium thiosulphate. If the amounts ofhydroxylamine and used iodine are figured from atomic weights, onemolecule of hydroxylamine salt requires two atoms of iodine. It fol-lows that one molecule of hydroxylamine is oxidized by two atoms ofiodine if the hydriodic acid formed is neutralized at once,2NH^0H+SIg=Np0+Hg0+4HI and proves accurately this formula by eudio-metric analysis of gases formed by the oxidation.The results obtained by this method were unsatisfactory.Iodine solution of whi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/hydroxylamine-its-quantitative-determination-and-some-double-salts-ne-solu-tion-and-titrate-back-with-sodium-thiosulphate-if-the-amounts-ofhydroxylamine-and-used-iodine-are-figured-from-atomic-weights-onemolecule-of-hydroxylamine-salt-requires-two-atoms-of-iodine-it-fol-lows-that-one-molecule-of-hydroxylamine-is-oxidized-by-two-atoms-ofiodine-if-the-hydriodic-acid-formed-is-neutralized-at-once2nh0hsig=np0hg04hi-and-proves-accurately-this-formula-by-eudio-metric-analysis-of-gases-formed-by-the-oxidationthe-results-obtained-by-this-method-were-unsatisfactoryiodine-solution-of-whi-image372159268.html
. Hydroxylamine : its quantitative determination and some double salts . ne solu-tion and titrate back with sodium thiosulphate. If the amounts ofhydroxylamine and used iodine are figured from atomic weights, onemolecule of hydroxylamine salt requires two atoms of iodine. It fol-lows that one molecule of hydroxylamine is oxidized by two atoms ofiodine if the hydriodic acid formed is neutralized at once,2NH^0H+SIg=Np0+Hg0+4HI and proves accurately this formula by eudio-metric analysis of gases formed by the oxidation.The results obtained by this method were unsatisfactory.Iodine solution of whi Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/hydroxylamine-its-quantitative-determination-and-some-double-salts-ne-solu-tion-and-titrate-back-with-sodium-thiosulphate-if-the-amounts-ofhydroxylamine-and-used-iodine-are-figured-from-atomic-weights-onemolecule-of-hydroxylamine-salt-requires-two-atoms-of-iodine-it-fol-lows-that-one-molecule-of-hydroxylamine-is-oxidized-by-two-atoms-ofiodine-if-the-hydriodic-acid-formed-is-neutralized-at-once2nh0hsig=np0hg04hi-and-proves-accurately-this-formula-by-eudio-metric-analysis-of-gases-formed-by-the-oxidationthe-results-obtained-by-this-method-were-unsatisfactoryiodine-solution-of-whi-image372159268.htmlRM2CHD8XC–. Hydroxylamine : its quantitative determination and some double salts . ne solu-tion and titrate back with sodium thiosulphate. If the amounts ofhydroxylamine and used iodine are figured from atomic weights, onemolecule of hydroxylamine salt requires two atoms of iodine. It fol-lows that one molecule of hydroxylamine is oxidized by two atoms ofiodine if the hydriodic acid formed is neutralized at once,2NH^0H+SIg=Np0+Hg0+4HI and proves accurately this formula by eudio-metric analysis of gases formed by the oxidation.The results obtained by this method were unsatisfactory.Iodine solution of whi
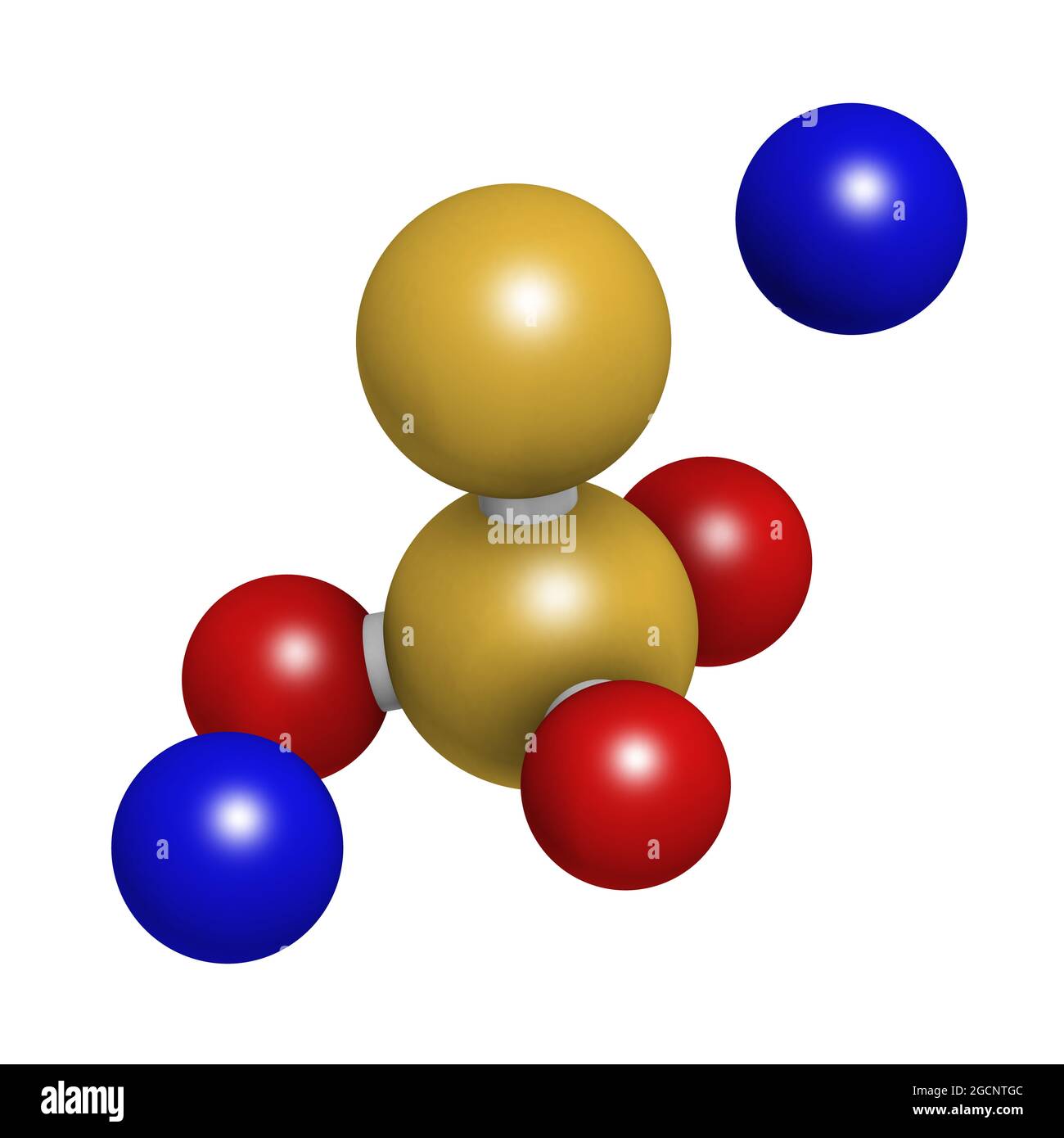 Sodium thiosulfate, chemical structure. 3D rendering. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-image438115340.html
Sodium thiosulfate, chemical structure. 3D rendering. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-image438115340.htmlRF2GCNTGC–Sodium thiosulfate, chemical structure. 3D rendering.
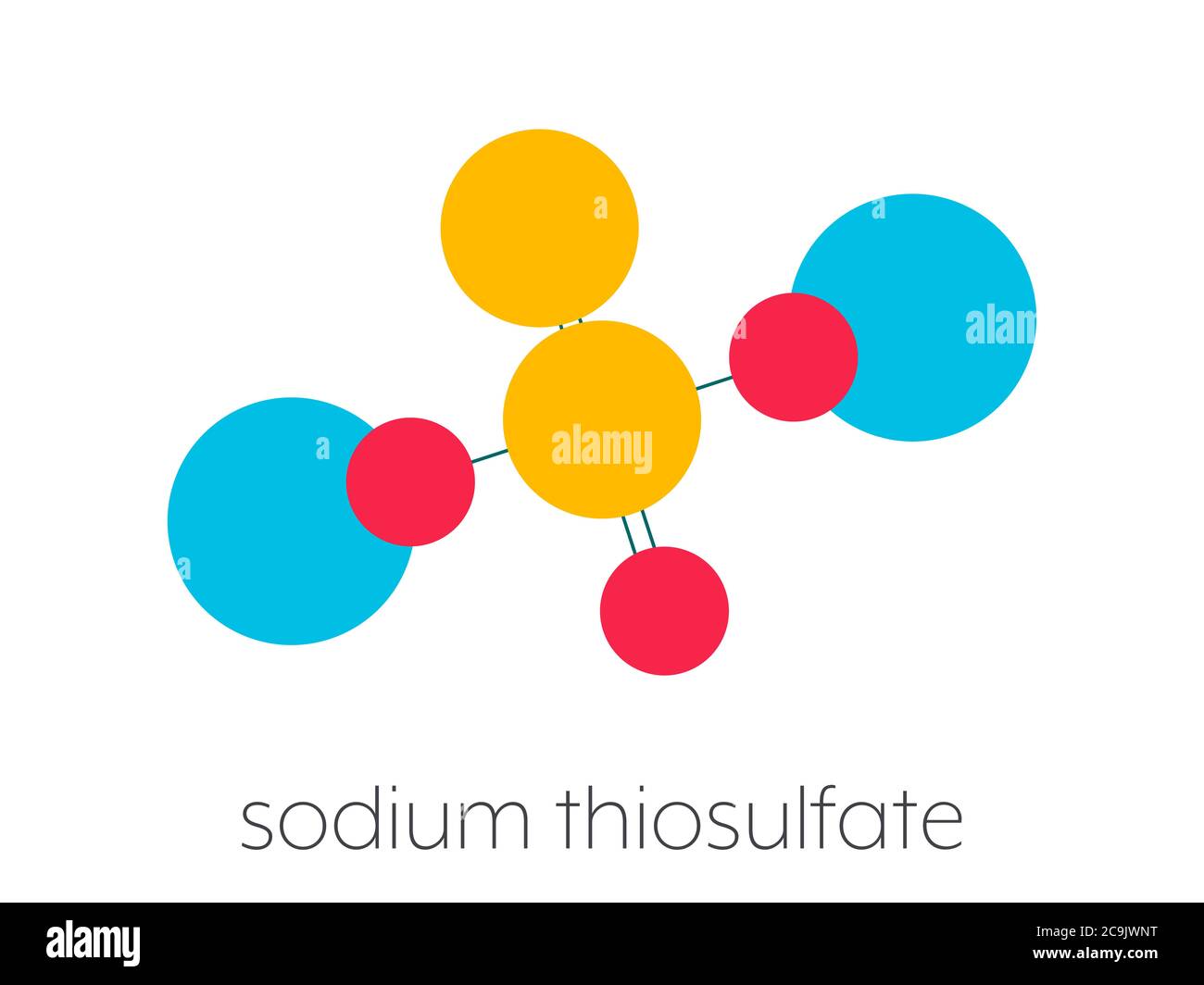 Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles conn Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-stylized-skeletal-formula-chemical-structure-atoms-are-shown-as-color-coded-circles-conn-image367364980.html
Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles conn Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-hyposulfite-salt-chemical-structure-stylized-skeletal-formula-chemical-structure-atoms-are-shown-as-color-coded-circles-conn-image367364980.htmlRF2C9JWNT–Sodium thiosulfate (hyposulfite) salt, chemical structure. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles conn
 Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563370759.html
Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563370759.htmlRF2RMFN47–Sodium thiosulfate or sodium thiosulphate on blue background.
 Opened plastic container of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-opened-plastic-container-of-sodium-thiosulphate-35119938.html
Opened plastic container of Sodium Thiosulphate Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-opened-plastic-container-of-sodium-thiosulphate-35119938.htmlRMC13RRE–Opened plastic container of Sodium Thiosulphate
 Pile of Sodium Thiosulphate Na2S2O3 colourless crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-pile-of-sodium-thiosulphate-na2s2o3-colourless-crystals-54655777.html
Pile of Sodium Thiosulphate Na2S2O3 colourless crystals Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/stock-photo-pile-of-sodium-thiosulphate-na2s2o3-colourless-crystals-54655777.htmlRFD4WP01–Pile of Sodium Thiosulphate Na2S2O3 colourless crystals
 Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412451.html
Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red), Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-represented-as-spheres-with-conventional-color-coding-sulfur-yellow-oxygen-red-image268412451.htmlRFWGK6PY–Sodium thiosulfate, chemical structure. 3D rendering. Atoms are represented as spheres with conventional color coding: sulfur (yellow), oxygen (red),
 . A treatise on pharmacy for students and pharmacists. Six-sided prism. Axes of the monoclinic system. Monoclinic double pyramid. Crystals of this system have three unequal axes, two being obliquelyinclined to each other, the other axis forming right angles with thesetwo. See Fig. 189. The fundamental forms of this system are the monoclinic doublepyramid or octohedron, Fig. 190, and the monoclinic prism, Fig. 191. Ferrous sulphate, borax, lead acetate, cupric acetate, tartaric acid,potassium chlorate and sodium acetate, sulphate, thiosulphate, phos-phate and carbonate, crystallize in this syst Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-treatise-on-pharmacy-for-students-and-pharmacists-six-sided-prism-axes-of-the-monoclinic-system-monoclinic-double-pyramid-crystals-of-this-system-have-three-unequal-axes-two-being-obliquelyinclined-to-each-other-the-other-axis-forming-right-angles-with-thesetwo-see-fig-189-the-fundamental-forms-of-this-system-are-the-monoclinic-doublepyramid-or-octohedron-fig-190-and-the-monoclinic-prism-fig-191-ferrous-sulphate-borax-lead-acetate-cupric-acetate-tartaric-acidpotassium-chlorate-and-sodium-acetate-sulphate-thiosulphate-phos-phate-and-carbonate-crystallize-in-this-syst-image370780627.html
. A treatise on pharmacy for students and pharmacists. Six-sided prism. Axes of the monoclinic system. Monoclinic double pyramid. Crystals of this system have three unequal axes, two being obliquelyinclined to each other, the other axis forming right angles with thesetwo. See Fig. 189. The fundamental forms of this system are the monoclinic doublepyramid or octohedron, Fig. 190, and the monoclinic prism, Fig. 191. Ferrous sulphate, borax, lead acetate, cupric acetate, tartaric acid,potassium chlorate and sodium acetate, sulphate, thiosulphate, phos-phate and carbonate, crystallize in this syst Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/a-treatise-on-pharmacy-for-students-and-pharmacists-six-sided-prism-axes-of-the-monoclinic-system-monoclinic-double-pyramid-crystals-of-this-system-have-three-unequal-axes-two-being-obliquelyinclined-to-each-other-the-other-axis-forming-right-angles-with-thesetwo-see-fig-189-the-fundamental-forms-of-this-system-are-the-monoclinic-doublepyramid-or-octohedron-fig-190-and-the-monoclinic-prism-fig-191-ferrous-sulphate-borax-lead-acetate-cupric-acetate-tartaric-acidpotassium-chlorate-and-sodium-acetate-sulphate-thiosulphate-phos-phate-and-carbonate-crystallize-in-this-syst-image370780627.htmlRM2CF6ED7–. A treatise on pharmacy for students and pharmacists. Six-sided prism. Axes of the monoclinic system. Monoclinic double pyramid. Crystals of this system have three unequal axes, two being obliquelyinclined to each other, the other axis forming right angles with thesetwo. See Fig. 189. The fundamental forms of this system are the monoclinic doublepyramid or octohedron, Fig. 190, and the monoclinic prism, Fig. 191. Ferrous sulphate, borax, lead acetate, cupric acetate, tartaric acid,potassium chlorate and sodium acetate, sulphate, thiosulphate, phos-phate and carbonate, crystallize in this syst
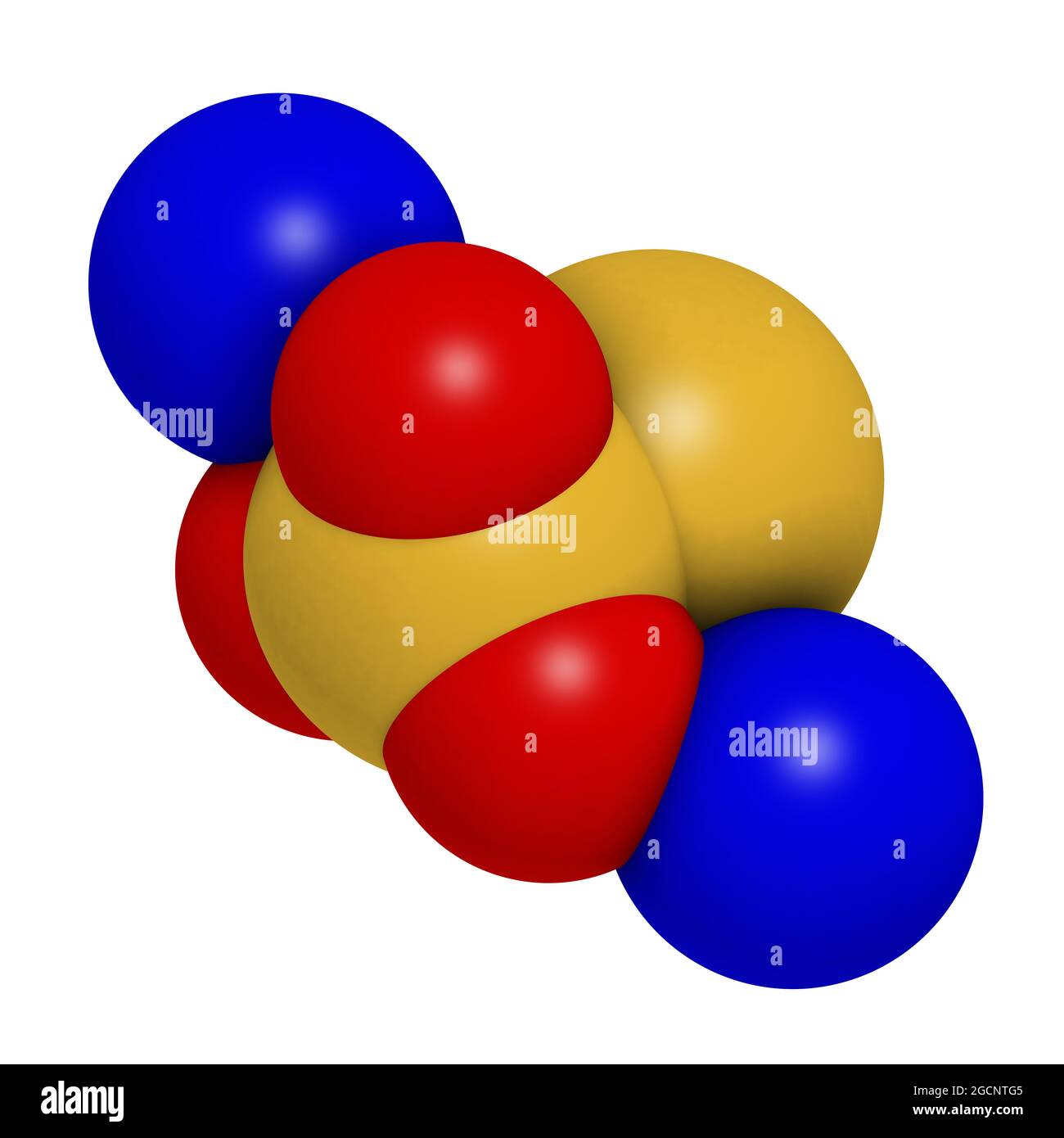 Sodium thiosulfate, chemical structure. 3D rendering. Atoms are Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-image438115333.html
Sodium thiosulfate, chemical structure. 3D rendering. Atoms are Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-chemical-structure-3d-rendering-atoms-are-image438115333.htmlRF2GCNTG5–Sodium thiosulfate, chemical structure. 3D rendering. Atoms are
 Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563369999.html
Sodium thiosulfate or sodium thiosulphate on blue background. Stock Photohttps://www.alamy.com/licenses-and-pricing/?v=1https://www.alamy.com/sodium-thiosulfate-or-sodium-thiosulphate-on-blue-background-image563369999.htmlRF2RMFM53–Sodium thiosulfate or sodium thiosulphate on blue background.
Search Results for Sodium thiosulphate Stock Photos and Images (47)
Page 1 of 1

